Pulmonary Embolism (PE): Symptoms, Diagnosis and Treatment
Sudden shortness of breath, chest pain, a racing heart — these symptoms could mean many things. But when a person has recently had surgery, a long flight, or has been confined to bed for a while, pulmonary embolism must be at the top of the differential. PE is the third most common acute cardiovascular emergency after heart attack and stroke — and one of the most underdiagnosed. Here's what causes it, what it looks like, how it is diagnosed, and how it is treated.
What Is Pulmonary Embolism and How Does It Develop
Pulmonary embolism is the acute blockage of the pulmonary artery or its branches by a clot — which usually formed not in the lungs but in the veins of the legs or pelvis. The clot detaches, travels through the bloodstream, passes through the right side of the heart, and lodges in a pulmonary vessel — cutting off blood supply to part of the lung.
The causes of clot formation are described by Virchow's triad — three factors that, alone or in combination, set the process in motion:
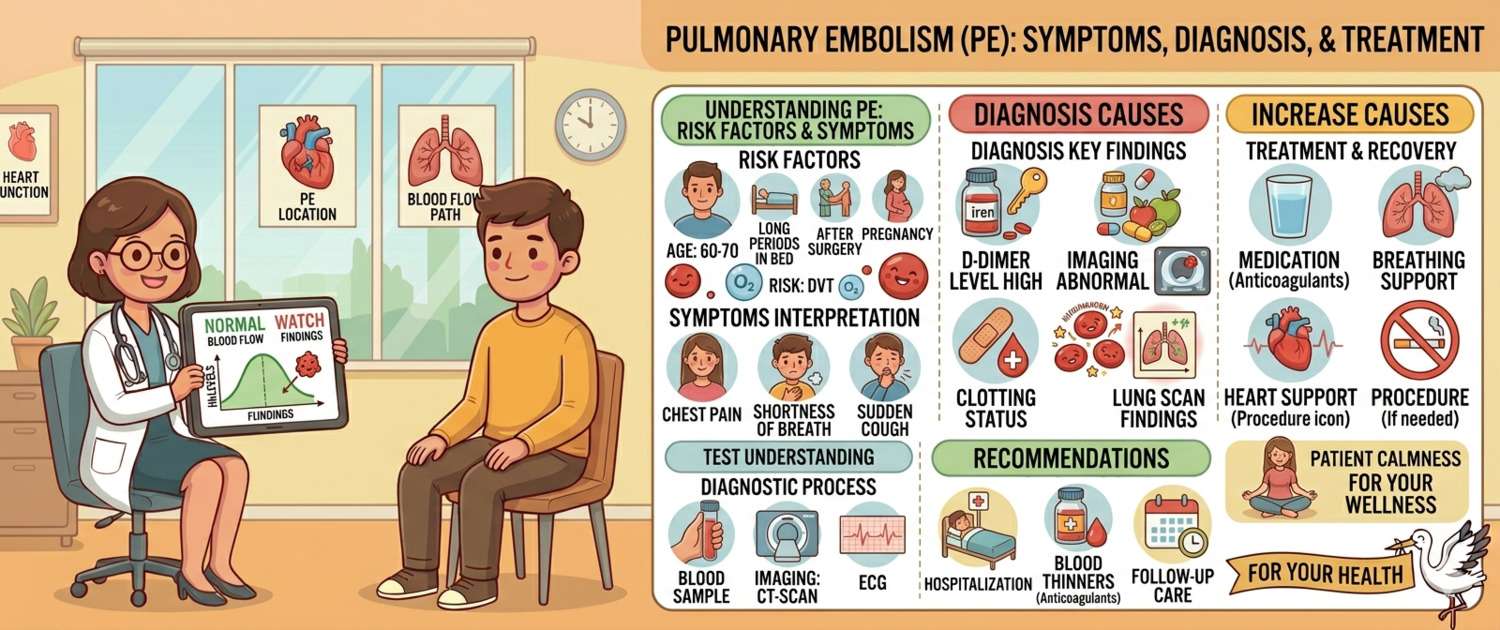
1. Slow blood flow — prolonged immobilisation: bed rest, a long flight or car journey, the post-operative period, limb paralysis.
2. Vessel wall damage — trauma, surgical procedures, venous inflammation (phlebitis), catheters and intravenous access.
3. Increased clotting tendency (hypercoagulability) — malignancy (especially pancreatic, lung, and gastric cancer), pregnancy and the postpartum period, hormone therapy and oral contraceptives, hereditary thrombophilia (protein C or S deficiency, Factor V Leiden mutation), antiphospholipid syndrome.
Imagine a river with sluggish current, a damaged bank, and thickened water — a clot in this environment is almost inevitable. PE is what happens when the resulting "blockage" drifts downstream and obstructs a critical channel.
Classification of PE by Severity
The severity of PE is determined by how severely right ventricular function is compromised — the chamber that pumps blood into the lungs.
Massive PE — blockage of the main trunk or large branches. Right ventricular pressure rises acutely and the chamber fails. Clinical picture: cardiogenic shock (systolic BP < 90 mmHg), loss of consciousness, cardiac arrest. Mortality without treatment: 30–50%.
Submassive PE — significant clot burden without shock, but with evidence of right ventricular strain on ECG, echocardiography, or elevated troponin. Moderate risk. Requires hospitalisation and active treatment.
Non-massive (small) PE — blockage of small branches, normal haemodynamics, no right ventricular strain. Low mortality with timely treatment.
Symptoms of PE: Why It Gets Missed
PE is treacherous because its symptoms are non-specific and are easily attributed to other conditions.
The classic PE triad:
- Dyspnoea — sudden, without obvious cause, worsening with exertion. The most common symptom (present in 80% of patients).
- Chest pain — usually pleuritic (worsens with breathing and coughing), less commonly substernal as in infarction. Present in 40–60%.
- Haemoptysis — blood in sputum. A sign of pulmonary infarction, present in only 10–15% — when a small branch has been occluded and lung tissue has died.
Other symptoms: tachycardia, anxiety and a sense of impending doom, sweating, cough, cyanosis (blue lips in massive PE), syncope.
Signs of deep vein thrombosis (DVT) — the clot source: pain, swelling, and redness in one leg (usually the calf or thigh) — present in only 25–30% of PE patients, because by then the clot has already left.
The principal diagnostic trap: PE symptoms are identical to pneumonia (dyspnoea, pleuritic pain, cough) and pericarditis (chest pain). This is exactly why instrumental confirmation is essential.
Diagnosing PE: From D-Dimer to CT Angiography
PE cannot be diagnosed from symptoms alone — laboratory and imaging investigations are required.
Clinical probability assessment — the first step. The doctor uses the Wells score or Geneva score: points for risk factors (recent surgery, prior DVT, tachycardia, malignancy) establish how likely PE is before any tests are ordered.
D-dimer — a fibrin breakdown product, a marker of active clot formation and dissolution. With low clinical probability, a normal D-dimer effectively rules out PE. The problem: D-dimer is extremely non-specific — it rises with any inflammation, in pregnancy, after surgery, and in malignancy. An elevated D-dimer does not confirm PE — it simply says "further investigation is needed."
CT pulmonary angiography (CTPA) — the gold standard. Contrast is injected intravenously and the scanner visualises the clot directly within the vessel. Sensitivity and specificity exceed 95%. With high clinical probability, CTPA is ordered directly, bypassing D-dimer.
ECG — changes in PE are often non-specific (tachycardia, right-sided changes, the S1Q3T3 pattern). Helps exclude myocardial infarction as the cause of symptoms.
Severity markers: troponin (rises with right ventricular overload — indicates severity and risk), BNP/NT-proBNP. A coagulation panel is important before starting anticoagulation and when thrombophilia is suspected.
A complete blood count excludes anaemia as a cause of dyspnoea and detects leucocytosis suggesting pneumonia (differential diagnosis). CRP helps assess the inflammatory component.
Echocardiography — in massive PE, right ventricular overload and a "D-shaped" interventricular septum are visible. In unstable patients who cannot undergo CTPA, echocardiography becomes the primary tool.
Treatment of PE
The goals of treatment are to arrest and dissolve the clot, restore pulmonary blood flow, and prevent recurrence.
Anticoagulation — the foundation of treatment for non-massive and submassive PE. Anticoagulants do not dissolve an existing clot but prevent it from growing and stop new ones forming:
- Direct oral anticoagulants (DOACs) — rivaroxaban, apixaban. First choice for most patients: fixed doses, no routine laboratory monitoring needed, suitable for outpatient treatment.
- Low-molecular-weight heparins (enoxaparin) — for hospitalised patients, pregnancy, and malignancy.
- Warfarin — for mechanical heart valves and long-term treatment. Requires regular INR monitoring (target INR 2.0–3.0) via a coagulation panel.
Duration: 3 months for provoked PE (after surgery); 6 months or longer for unprovoked PE; indefinite for recurrent or malignancy-associated PE.
Thrombolytic therapy (thrombolysis) — intravenous agents that dissolve the clot (alteplase). Indicated for massive PE with cardiogenic shock. Rapidly restores blood flow but carries a high bleeding risk, including intracranial haemorrhage. Used only in an intensive care setting.
Surgical treatment — embolectomy (clot removal) or catheter-directed fragmentation when thrombolysis is contraindicated.
Oxygen therapy and supportive care — for all forms of PE.
When to Call Emergency Services
Call an ambulance immediately for:
- Sudden dyspnoea at rest — especially after surgery, prolonged immobilisation, or a long flight.
- Chest pain that worsens with breathing.
- Coughing up blood.
- Loss of consciousness or sudden severe drop in blood pressure.
- Rapid heartbeat combined with dyspnoea and leg pain or swelling.
Any of these in isolation does not necessarily mean PE. Their combination in the presence of risk factors is sufficient reason to call emergency services without waiting.
Preventing PE After Surgery and During Travel
Most cases of PE are preventable. Key measures:
In hospital: compression stockings and pneumatic calf compression during immobilisation; prophylactic heparin or DOAC dosing in high-risk surgical patients; early mobilisation after surgery.
During travel: stand and walk for a few minutes every two hours on long flights (> 4 hours); wear compression stockings; stay hydrated.
Long-term: treating varicose vein disease; managing malignancy; discontinuing oral contraceptives when thrombophilia is identified; prolonged anticoagulation for recurrent thrombosis.
Conclusion
Pulmonary embolism is an emergency that can be missed — and prevented. Its danger lies in symptoms that mimic pneumonia or heart attack. The key to diagnosis is clinical thinking: with any sudden dyspnoea against a background of risk factors, PE must be on the differential. The key to treatment is rapid anticoagulation, which when started promptly reduces mortality five to eight times.
This article is for informational purposes only. Interpreting test results and prescribing treatment is exclusively the responsibility of a physician.
Frequently Asked Questions
Pulmonary embolism is an acute blockage of a pulmonary artery by a clot that has typically detached from the veins of the legs or pelvis. The danger lies in abrupt right ventricular overload: massive PE causes cardiogenic shock and possible cardiac arrest. Mortality without treatment in massive PE reaches 30–50%. With timely anticoagulation, the prognosis is significantly better.
The classic symptoms are sudden dyspnoea (especially at rest), chest pain that worsens with breathing, and tachycardia. Haemoptysis is only present in 10–15% of cases. Swelling and pain in one leg — a sign of the clot source — is found in only about a third of PE patients, as the clot has usually already left. The deceptiveness of PE is that symptoms are easily confused with pneumonia or myocardial infarction. With risk factors present and sudden dyspnoea — call emergency services immediately.
The primary laboratory marker is D-dimer: a normal result with low clinical probability effectively rules out PE. For severity assessment, troponin is ordered — its elevation indicates right ventricular overload. A coagulation panel is needed before starting anticoagulants. A complete blood count and CRP help differentiate PE from pneumonia. The gold standard for visualisation is CT pulmonary angiography.
Anticoagulation is the foundation: it prevents the clot from growing and stops new ones forming. For non-massive and submassive PE, direct oral anticoagulants (rivaroxaban, apixaban) or low-molecular-weight heparins are used. For massive PE with shock — thrombolysis (clot dissolution with alteplase). Treatment duration ranges from 3 months to lifelong, depending on the cause and recurrence risk.
Post-surgical PE prevention includes early mobilisation (walking the day after surgery), compression stockings, pneumatic calf compression during and after the procedure, and prophylactic anticoagulants — usually low-molecular-weight heparin or rivaroxaban — in high-risk patients. Dehydration and prolonged immobility are the main additional risk factors to avoid.
Flying is possible after PE but with restrictions. For the first 4–6 weeks after the acute event, long flights are not recommended — recurrence risk is high. When travel is necessary: continue anticoagulation, wear compression stockings, and walk for a few minutes every two hours. Specific recommendations depend on the severity of the PE episode and are determined by the treating physician.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service