Tumour Marker Panel: What's Included, Normal Levels and Results
You received a "tumour marker panel" result showing a value slightly above the reference range. First thought — cancer? Stop. Tumour markers are among the most overestimated and misunderstood tools in laboratory medicine. Most of them are not designed to screen for cancer in healthy people, rise with many benign conditions, and almost never establish a diagnosis on their own. Here is what tumour markers actually are, what they genuinely show, and in which context their results carry meaning.
What's Included in a Tumour Marker Panel and How It Works
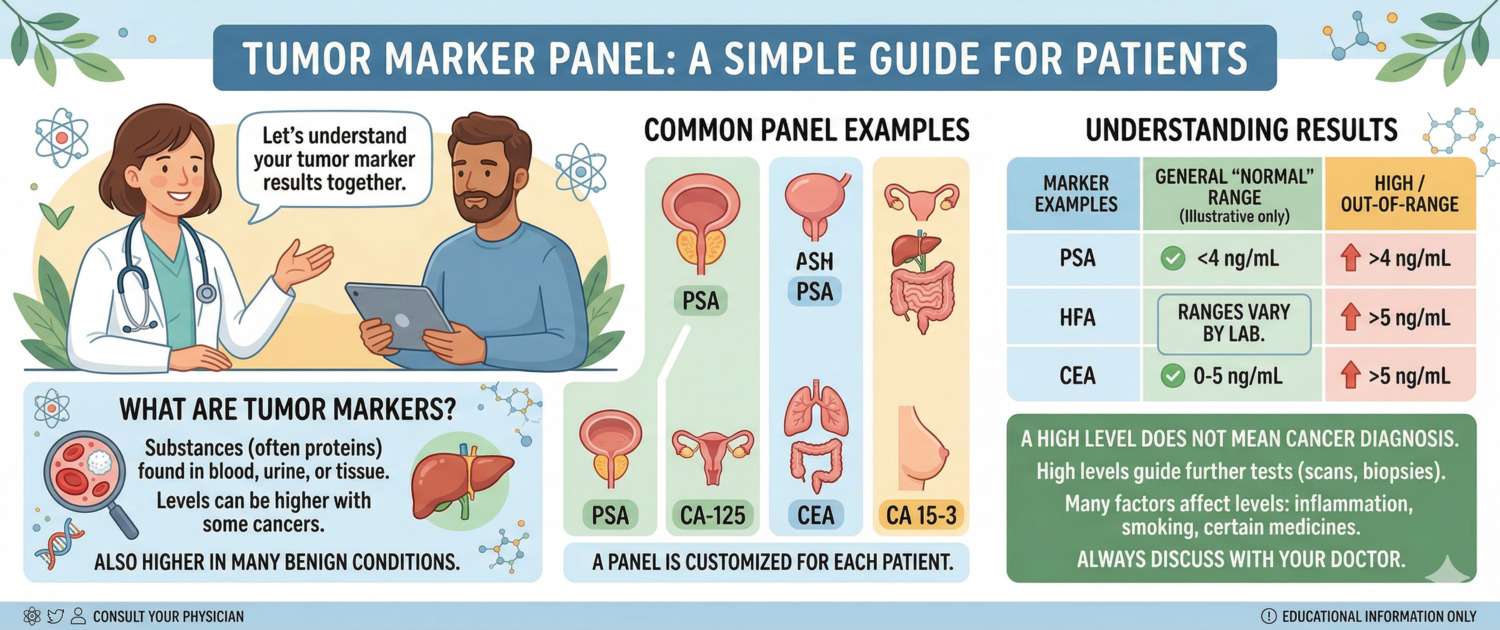
Tumour markers are substances — proteins, hormones, antigens — whose concentration in blood, urine or other body fluids changes in association with malignant tumours. They may be produced by tumour cells themselves or by normal cells responding to the presence of a tumour.
The fundamental problem: most tumour markers are not specific to cancer. The same substance can rise with inflammation, benign tumours, liver cirrhosis, pregnancy, or physical exertion. This means:
- Elevated marker ≠ cancer — in most cases the cause is something else
- Normal marker ≠ no cancer — many tumours produce no markers at all
- Diagnostic value is greatest for monitoring an already established diagnosis and tracking treatment response
The exception — a handful of markers with high organ and tumour specificity, most notably PSA for prostate cancer and hCG for germ cell tumours.
Why Tumour Markers Are Ordered: Screening and Monitoring
Tumour markers are used in three fundamentally different contexts, each with different diagnostic value.
Treatment monitoring — the most evidence-based application. After surgery, chemotherapy or radiotherapy, a falling marker confirms treatment response. A rising marker during remission is an early sign of relapse, often preceding clinical symptoms by weeks or months. This is precisely why tumour markers are incorporated into follow-up protocols for most oncological diagnoses.
Diagnosis in symptomatic patients — when there is a well-founded clinical suspicion of a specific tumour, a marker can help clarify the diagnosis or guide the investigation pathway. For example, an elevated PSA in a man with urological symptoms warrants prostate biopsy.
Screening in healthy people — the least evidence-based application for most markers. The exception: PSA is used for prostate cancer screening in men over 45–50 when the patient understands and accepts the method's limitations.
Key Tumour Markers and What They Show
PSA (prostate-specific antigen)
The most extensively studied and clinically significant tumour marker. Produced by prostate cells — both normal and malignant. Elevated in prostate cancer, benign prostatic hyperplasia, and prostatitis. Age-adjusted normal values: below 50 years < 2.5 ng/mL; 50–60 years < 3.5 ng/mL; over 60 < 4.5–6.5 ng/mL. Full details in the PSA blood test article.
hCG (human chorionic gonadotropin) and β-hCG
Physiologically produced during pregnancy — that is its primary function. As a tumour marker: elevated in non-seminomatous germ cell tumours of the testis, choriocarcinoma of the uterus, and some gastrointestinal and lung malignancies. In men and non-pregnant women, any detectable hCG level requires oncological evaluation. After treatment for germ cell tumours, hCG is used to monitor remission — its rise is the first signal of relapse.
AFP (alpha-fetoprotein)
Synthesised by the fetal liver and yolk sac — in healthy adults, levels are minimal (< 10 IU/mL). Elevated in hepatocellular carcinoma, non-seminomatous germ cell tumours, and occasionally in liver metastases from other primaries. Moderate elevation is seen in cirrhosis and viral hepatitis without malignancy. AFP is used alongside hCG in staging and monitoring germ cell tumours.
CEA (carcinoembryonic antigen)
One of the oldest tumour markers. Elevated in colorectal, lung, breast, and pancreatic cancers. Normal < 5 ng/mL (< 10 ng/mL in smokers). Moderate elevation occurs in inflammatory bowel disease, hepatitis, cirrhosis, and smoking — without cancer. Its primary clinical role is monitoring colorectal cancer recurrence after surgery.
CA-125
The primary marker for ovarian cancer. Normal < 35 IU/mL. Elevated in ovarian, endometrial, and fallopian tube cancers. Also rises with endometriosis, uterine fibroids, pelvic inflammatory disease, and even during menstruation — which substantially limits its value as a standalone screening test. It is combined with pelvic ultrasound to improve diagnostic accuracy.
CA-19-9
Marker for pancreatic, biliary tract, and occasionally gastric and colorectal cancers. Normal < 37 IU/mL. Rises markedly in obstructive jaundice of any cause — cholelithiasis, cholangitis — without malignancy. Used primarily to monitor pancreatic cancer and assess resectability.
Specific markers for haematological malignancies
In multiple myeloma, the key markers are M-protein (paraprotein) on serum protein electrophoresis, serum free light chains, and β₂-microglobulin — the last of which is incorporated in the ISS staging system. In lymphomas, LDH rises as a tumour burden marker. When a haematological malignancy is suspected, a complete blood count with full differential is essential.
Tumour Marker Normal Ranges: An Important Caveat
Each tumour marker has its own reference interval established in a healthy population. Exceeding the upper limit does not mean a tumour is present — it means the result requires interpretation in clinical context.
| Marker | Approximate normal | Main tumour associations |
|---|---|---|
| Total PSA | < 4 ng/mL (age-adjusted) | Prostate cancer |
| β-hCG | < 5 mIU/mL (non-pregnant / men) | Germ cell tumours, choriocarcinoma |
| AFP | < 10 IU/mL | Liver cancer, germ cell tumours |
| CEA | < 5 ng/mL | Colorectal, lung cancer |
| CA-125 | < 35 IU/mL | Ovarian cancer |
| CA-19-9 | < 37 IU/mL | Pancreatic cancer |
Reference ranges vary slightly by method and laboratory — always compare your result against the range printed on your own report.
Elevated Tumour Markers: Causes and Results Interpretation
The degree of elevation matters. A mild excess of 1.5–2 times the upper limit in the absence of symptoms is more likely a false-positive. A 5–10-fold or greater elevation alongside symptoms is a substantial argument for further investigation.
Approach to an elevated tumour marker:
- Exclude physiological and benign causes (inflammation, pregnancy, cirrhosis)
- Repeat the test in 4–6 weeks — sustained elevation is more significant than a single result
- Order targeted imaging (ultrasound, CT, MRI) of the relevant anatomical region
- Interpret the result together with the clinical picture
Self-interpreting tumour markers without a physician carries a high risk of both over-diagnosis and missed genuine pathology.
How to Prepare for Tumour Marker Testing
Fast for at least 8 hours before the blood draw. For PSA — 2–3 days before the test: avoid sexual activity, cycling, and rectal procedures — all transiently elevate PSA. Draw blood before any biopsy or invasive procedure — post-procedural marker elevation can persist for weeks. Declare all medications: finasteride and dutasteride halve PSA; some chemotherapy agents affect other markers.
For serial monitoring, always use the same laboratory and the same assay method — inter-laboratory calibration differences can mimic genuine marker changes.
When to Seek Urgent Medical Attention
Contact an oncologist or the relevant specialist promptly when: PSA exceeds 20 ng/mL on a first measurement; hCG is detectable above 100 mIU/mL in a man or non-pregnant woman; AFP exceeds 400 IU/mL alongside a newly discovered liver lesion; a marker is rising during remission in a patient with an established cancer diagnosis — possible relapse.
This article is for informational purposes only. Test ordering, result interpretation and investigation strategy are determined by an oncologist or relevant specialist.
Frequently Asked Questions
No — an elevated tumour marker is not a cancer diagnosis. Most markers rise with inflammatory disease, benign tumours, liver cirrhosis, pregnancy and other conditions. For example, a moderately elevated PSA is more often caused by benign prostatic hyperplasia or prostatitis than by cancer. Every result requires physician interpretation alongside clinical findings and imaging.
Most tumour markers are not recommended for screening healthy people due to low specificity — the high rate of false-positive results generates anxiety and leads to unnecessary investigations without genuine benefit. The exception is PSA for men over 45–50: its use in prostate cancer screening is justified when discussed and agreed with a physician. Tumour markers are most valuable for monitoring established diagnoses.
The primary ovarian cancer marker is CA-125 (normal < 35 IU/mL). However, it also rises with benign gynaecological conditions: endometriosis, fibroids, and pelvic inflammatory disease. This is why CA-125 is not used as a standalone screening test — it is combined with transvaginal ultrasound. In patients with confirmed ovarian cancer, CA-125 is a key tool for monitoring treatment response.
In pregnancy, hCG is the normal placental hormone. In non-pregnant women and men, any detectable hCG level is an oncological alert: it is produced by germ cell tumours of the testis, choriocarcinoma of the uterus, and occasionally gastrointestinal malignancies. After treatment, hCG trends are used to monitor remission. In multiple myeloma, hCG is sometimes mildly elevated due to cross-reactivity with other glycoprotein hormones.
Frequency depends on the specific disease and the follow-up protocol set by the oncologist. Typically during active treatment — before each cycle or every 1–3 months. During remission — every 3–6 months for the first 2–3 years, then less frequently. A stable or declining marker is reassuring; a rising marker prompts unscheduled imaging.
Yes — a complete blood count adds important information to a tumour marker panel: anaemia, thrombocytopenia or changes in the white cell differential may indicate bone marrow involvement or displacement of normal haematopoiesis by tumour. In haematological malignancies such as lymphoma and myeloma, a full blood count with differential is one of the primary diagnostic tools.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service