Neutrophils in Blood: Normal Levels, Causes and Significance
Neutrophils are the first cells to rush toward a site of infection. They make up approximately 50–70% of all white blood cells and serve as the primary instrument of rapid antibacterial defense: reaching the site of inflammation within 30–60 minutes, engulfing and destroying bacteria through phagocytosis, and forming purulent exudate. This is why any leukocyte differential analysis begins with neutrophils — their deviation in either direction carries the most immediate diagnostic information.
What Neutrophils Are and How They Are Classified
Neutrophils are granulocytic leukocytes that mature in the bone marrow from myeloblasts through several intermediate stages. Mature cells enter the bloodstream and circulate for 6–12 hours before migrating into tissues, where they survive for another 1–2 days.
In the blood differential, neutrophils are divided into two subsets by nuclear shape:
Segmented neutrophils (segs) — mature, fully functional cells. The nucleus is divided into 2–5 lobes connected by thin strands. They constitute the vast majority of circulating neutrophils in health (47–72%).
Band neutrophils — immature cells with an unsegmented, rod-shaped or horseshoe-shaped nucleus. Normally few in number (1–6%). A significant increase in bands — the "left shift" — signals that the bone marrow is urgently releasing immature cells in response to acute infection or inflammation.
In severe infections, even more immature precursors — metamyelocytes and myelocytes — may appear in the blood, indicating extreme hematopoietic stress.
Neutrophil granules contain powerful bactericidal substances: myeloperoxidase, lysozyme, defensins, and lactoferrin. After engulfing a bacterium, the neutrophil destroys it through a "respiratory burst" — a sudden release of reactive oxygen species — and then self-destructs, becoming part of the pus.
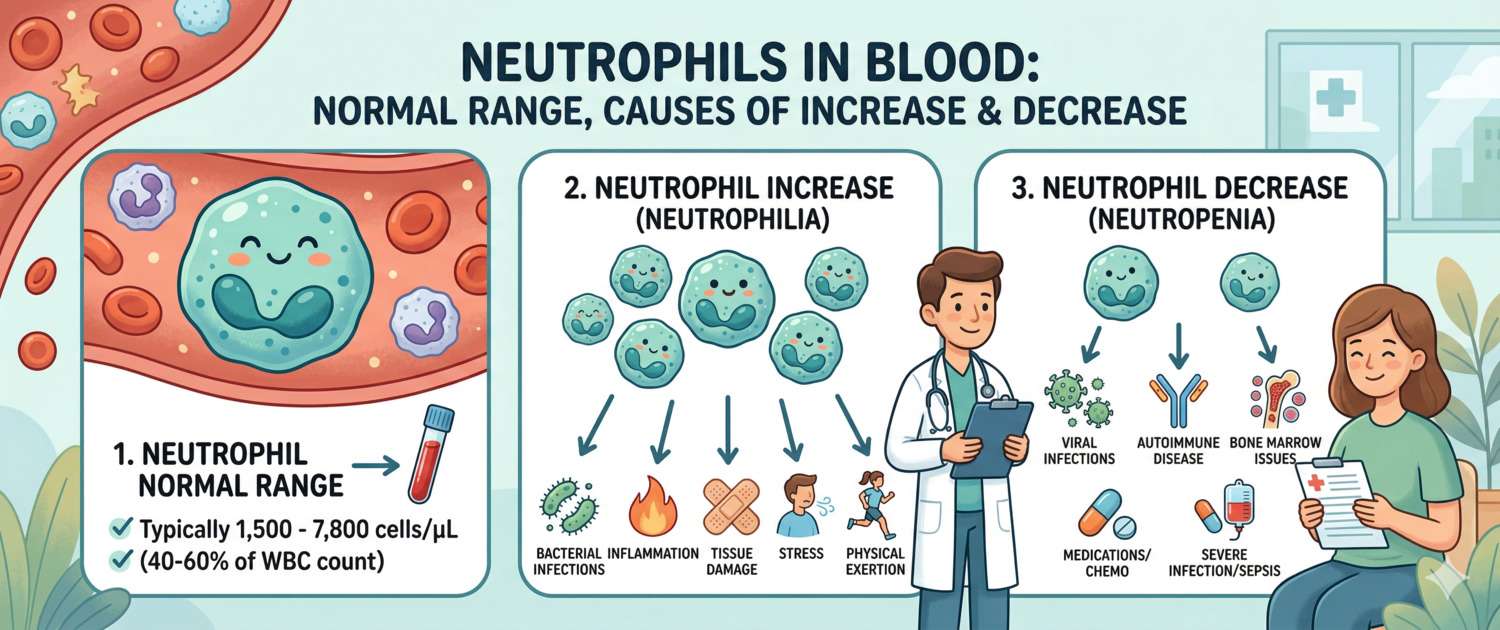
Normal Neutrophil Levels by Age
Reference values differ substantially across childhood. A key feature: in newborns neutrophils predominate; their proportion then falls and by age 4–6 equalizes with lymphocytes — the "second physiological crossover." Adult proportions are not reached until age 10–12.
| Age | Segmented neutrophils (%) | Band neutrophils (%) | Absolute neutrophils (×10⁹/L) |
|---|---|---|---|
| Newborns | 47–70 | 3–12 | 6.0–26.0 |
| 1 month – 1 year | 15–45 | 1–5 | 1.5–8.5 |
| 1–4 years | 25–55 | 1–5 | 1.5–8.5 |
| 4–6 years | 35–65 | 1–5 | 1.5–8.0 |
| 6–12 years | 40–60 | 1–5 | 1.8–8.0 |
| 12–18 years | 40–65 | 1–5 | 1.8–7.5 |
| Adults | 47–72 | 1–6 | 1.8–7.5 |
The absolute neutrophil count (ANC) is the most clinically important parameter, especially for infection risk assessment and in oncology patients:
- ANC > 1.5 × 10⁹/L — normal
- ANC 1.0–1.5 × 10⁹/L — mild neutropenia
- ANC 0.5–1.0 × 10⁹/L — moderate neutropenia
- ANC < 0.5 × 10⁹/L — severe neutropenia; high risk of life-threatening infection
During pregnancy, neutrophils physiologically rise — especially in the third trimester and postpartum. Mild neutrophilia in pregnancy is normal and requires no additional investigation.
How to Prepare for a Neutrophil Count
Neutrophils are measured as part of a complete blood count with differential — there is no separate test.
- Blood is drawn from a vein in the morning, fasting or 3–4 hours after a light meal
- Physical exercise — even walking — transiently elevates neutrophils by mobilizing the "marginal pool" (cells adhering to vessel walls). Levels can rise 50–100% within 30 minutes. Avoid intense activity for 24 hours beforehand and rest quietly for 15–20 minutes before the draw
- Smoking chronically elevates neutrophils — in smokers the upper normal limit is effectively higher
- Stress (physical or emotional) raises neutrophils via an adrenaline mechanism
- For serial monitoring: same laboratory, same conditions each time
The differential must always be interpreted using absolute values, not percentages alone: 70% neutrophils with total leukocytes of 3 × 10⁹/L is neutropenia despite the apparently "normal" percentage.
Causes of High Neutrophils (Neutrophilia)
Neutrophilia — ANC > 7.5 × 10⁹/L in adults — is the most common finding in infections and inflammatory conditions.
| Cause | Degree of elevation | Characteristic features |
|---|---|---|
| Acute bacterial infection | Moderate–significant | Left shift; elevated CRP |
| Purulent inflammation (abscess, pneumonia) | Significant | Toxic granulation of neutrophils |
| Acute myocardial infarction | Moderate | First 12–24 hours |
| Chronic inflammation (flare) | Mild–moderate | Moderately elevated CRP |
| Stress, physical exercise | Mild, transient | Normalizes within hours |
| Corticosteroids | Moderate | Absent left shift |
| Pregnancy (3rd trimester) | Mild | Physiological |
| Smoking | Mild, chronic | Stable elevation |
| Malignancies | Variable | Often without left shift |
| Myeloproliferative disorders (CML) | Very high | All precursors in differential |
Glucocorticoids (prednisolone, dexamethasone) produce a characteristic "steroid neutrophilia": they mobilize neutrophils from the marginal pool and delay their tissue egress. Critically: there is no left shift and no toxic granulation — signs of true infection. Distinguishing steroid neutrophilia from infectious requires the clinical picture and ESR.
In chronic myeloid leukemia (CML), neutrophils may reach 50–200 × 10⁹/L with all maturation stages present — from myeloblasts to mature segmented cells. This is a leukemoid reaction or hematological malignancy — urgent hematology referral is required.
Causes of Low Neutrophils (Neutropenia)
Neutropenia — ANC < 1.5 × 10⁹/L — substantially increases the risk of bacterial and fungal infections. At ANC < 0.5 × 10⁹/L, the risk becomes life-threatening.
| Cause | Mechanism | Characteristic features |
|---|---|---|
| Viral infections (URTI, influenza, EBV) | Suppressed granulopoiesis + redistribution | Elevated lymphocytes |
| Chemotherapy and radiotherapy | Direct bone marrow suppression | Nadir at days 7–14 |
| Aplastic anemia | Stem cell failure | Pancytopenia |
| Autoimmune neutropenia | Anti-neutrophil antibodies | Isolated neutropenia |
| Drug-induced neutropenia | Immune-mediated or direct toxicity | Clear link to drug initiation |
| Cyclic neutropenia | Periodic suppression every 21 days | Recurrent infections |
| Severe B12 and folate deficiency | Ineffective granulopoiesis | Hypersegmented neutrophils |
| Felty's syndrome (RA + neutropenia) | Splenic sequestration | In rheumatoid arthritis context |
| HIV infection | Direct suppression + autoantibodies | Combined with lymphopenia |
Drug-induced neutropenia is a serious, frequently underrecognized problem. The most common agents: antithyroid drugs (methimazole, propylthiouracil) — require CBC monitoring every 2–4 weeks; clozapine — life-threatening agranulocytosis in approximately 1% of patients; certain antibiotics (chloramphenicol, co-trimoxazole), NSAIDs, carbamazepine.
Infectious mononucleosis (Epstein-Barr virus) frequently causes transient neutropenia combined with atypical lymphocytosis — a diagnostically significant combination.
Neutrophil Left and Right Shift
Neutrophil interpretation is incomplete without assessing the "differential shift" — the change in the ratio of mature to immature forms.
Left shift — increase in band neutrophils (> 6%) and/or appearance of metamyelocytes and myelocytes in circulation. Signals that the bone marrow has accelerated neutrophil production and released immature cells prematurely.
Causes: acute bacterial infection (most common), sepsis, severe inflammation, burns and major trauma, myeloproliferative disorders.
Degrees of left shift:
- Mild: bands 6–10%, no immature precursors
- Moderate: bands 10–20%, occasional metamyelocytes
- Marked: bands > 20%, metamyelocytes, myelocytes present — severe infectious or septic process
Right shift — reduction in band neutrophils with increased proportion of hypersegmented forms (> 5 lobes). Signals that neutrophils are "aging" — the bone marrow is producing them slowly or they are being retained in the circulation.
Causes: vitamin B12 and folate deficiency (classic — hypersegmented neutrophils in megaloblastic anemia); chronic kidney failure; hydroxyurea treatment.
Toxic granulation and Döhle bodies — morphological changes in neutrophils during severe infection and sepsis: intensified primary granulation (visible on microscopy of a stained smear) and pale blue-grey cytoplasmic inclusions. These findings amplify the diagnostic significance of neutrophilia when assessing severity.
When Neutrophil Abnormalities Require Medical Attention
Neutrophil interpretation is always multidimensional: absolute count + percentage + shift + clinical picture.
Scheduled visit to a doctor when:
- ANC < 1.5 × 10⁹/L on repeat testing — identify the cause of neutropenia
- Neutrophilia > 10 × 10⁹/L without an obvious explanation (no infection, no stress, no corticosteroids)
- Mild left shift with no symptoms — rule out a hidden infection source
Seek urgent care when:
- ANC < 0.5 × 10⁹/L — agranulocytosis; any sign of infection (temperature > 38°C) in this setting is a medical emergency
- Neutrophils > 30–50 × 10⁹/L with immature precursors present — leukemia must be excluded
- Patient taking antithyroid drugs or clozapine with neutrophils < 1.0 × 10⁹/L — immediate drug discontinuation and hematology referral
- Fever in a patient post-chemotherapy — febrile neutropenia — an infectious emergency regardless of the exact neutrophil level
Febrile neutropenia — temperature > 38°C with ANC < 0.5 × 10⁹/L — requires hospitalization and immediate broad-spectrum antibiotic therapy while awaiting culture results. Any delay is measured in hours, not days.
This article is for informational purposes only and does not replace professional medical advice. Consult a GP or hematologist if your neutrophil count is outside the normal range.
Frequently Asked Questions
Both are neutrophils but at different stages of maturity. Band neutrophils are immature: their nucleus has not yet divided into lobes and appears as a horseshoe or rod shape. Segmented neutrophils are mature and functionally more active: the nucleus is divided into 2–5 lobes. In health, segmented forms predominate (47–72%) and bands are few (1–6%). An increase in bands above 6% is called a left shift — a signal that acute infection or severe inflammation has triggered accelerated release of immature cells from the bone marrow.
Yes, and this is common. Neutrophilia without infection occurs with: physical exercise (transient), emotional or physical stress via an adrenaline surge, corticosteroid use, smoking, third-trimester pregnancy, acute myocardial infarction in the first 24 hours, and chronic inflammation of any cause. Distinguishing infectious from non-infectious neutrophilia relies on the combination with a left shift and elevated CRP — steroid or stress-related neutrophilia typically lacks both these features.
Neutropenia during a viral infection is a normal immune response, not a pathology. Viruses suppress bone marrow granulopoiesis and trigger neutrophil redistribution into tissues. Simultaneously, lymphocytes rise — the antiviral immune response is lymphocyte-driven. Mild transient neutropenia with a viral illness typically resolves spontaneously within 1–2 weeks. Neutropenia below 1.0 × 10⁹/L persisting more than 3–4 weeks after recovery warrants further investigation.
Very dangerous. At ANC < 0.5 × 10⁹/L (severe neutropenia), bacterial and fungal defenses are critically impaired: any infection can rapidly progress to sepsis. The neutrophil nadir with most chemotherapy regimens falls at days 7–14 after administration. Fever > 38°C during severe neutropenia — febrile neutropenia — requires emergency hospitalization and immediate antibiotics. Recovery is monitored alongside monocytes: rising monocytes after the nadir is the first laboratory sign of bone marrow recovery.
It depends on the degree and context. Neutrophilia up to 10 × 10⁹/L in a smoker, after exercise, or following stress typically requires no active workup. Neutrophilia > 10 × 10⁹/L without an obvious explanation — especially if accompanied by a left shift — is a reason for a scheduled GP visit to rule out occult infection, chronic inflammation, and at very high values (> 30 × 10⁹/L), myeloproliferative disease.
Both markers rise in bacterial infection but at different speeds. Neutrophilia with a left shift is an early marker: it develops within 4–6 hours of infection onset. ESR is a lagging marker: it rises after 24–48 hours and normalizes much more slowly (weeks). In an acute severe infection, neutrophils may be markedly elevated while ESR is only just beginning to climb — this is expected, not a contradiction. In chronic inflammation, ESR can remain persistently elevated with only moderate neutrophilia.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service