Electrolyte Panel: What's Included, Normal Values and Results
Night cramps in the calves, unexplained palpitations, sudden profound weakness after a bout of vomiting or diarrhoea — behind all of these symptoms, electrolyte imbalance is a common culprit. Electrolytes are charged mineral ions that govern the electrical activity of every cell in the body: from the nerve impulse to the heartbeat. An electrolyte panel is one of the core biochemical tests used to assess this balance from a single blood draw. This article covers what the panel includes, how to prepare, what counts as normal, and what each deviation means.
What Are Electrolytes and Why Is the Panel Needed?
Electrolytes are ions dissolved in blood and interstitial fluid: sodium, potassium, chloride, bicarbonate, calcium, magnesium, and phosphorus. They perform several critically important functions simultaneously.
First, they maintain osmotic pressure — regulating how much water sits inside cells versus outside. Think of a cell as a bag with a semi-permeable wall: the concentration of electrolytes on each side determines whether water flows in or out. Disrupt this balance and cells either swell or shrink.
Second, electrolytes generate the electrochemical gradients that underpin nerve and muscle function. Every nerve impulse is a wave of sodium and potassium redistribution across the neuron membrane. Every heartbeat is the result of precisely coordinated ion flows. Even a small deviation from normal can trigger an arrhythmia or muscle cramp.
Third, electrolytes maintain acid-base equilibrium — blood pH must remain within the narrow range of 7.35–7.45. Bicarbonate and chloride are the primary buffering systems of this balance.
The kidneys are the central regulators of electrolyte balance: they filter the entire blood volume approximately 50 times per day, reabsorbing what is needed and excreting what is not. This is why any serious impairment of kidney function is immediately reflected in the electrolyte panel. For a comprehensive renal assessment, the electrolyte panel is ordered alongside the kidney function test.
When Is an Electrolyte Panel Ordered?
Indications span a wide range of clinical situations — from emergencies to routine monitoring.
Acute conditions where the panel is mandatory:
- Severe vomiting or diarrhoea — electrolytes are lost with fluid at a critical rate
- Dehydration of any degree
- Cardiac arrhythmia of unknown cause
- Muscle cramps, weakness, or paralysis
- Altered consciousness or confusion
- Diuretic overdose or toxicity
Routine and preventive monitoring:
- Chronic kidney disease — electrolytes checked at every visit
- Diabetes mellitus — particularly during decompensation and diabetic ketoacidosis
- Long-term use of diuretics, ACE inhibitors, angiotensin receptor blockers, or digoxin
- Arterial hypertension — monitoring the effect of antihypertensive therapy
- Adrenal insufficiency and primary hyperaldosteronism
- Eating disorders, anorexia, or refeeding syndrome
- Pregnancy — screening each trimester when complications are present
How to Prepare and Get Tested
Serum electrolytes are relatively stable, but several factors can significantly distort the result.
Preparation guidelines:
- Blood is drawn fasting — at least 8–12 hours after the last meal. This matters most for potassium: a meal rich in potassium (bananas, potatoes, nuts) the evening before can transiently raise the level.
- Avoid alcohol and intense physical exercise for 24 hours: strenuous training causes a temporary shift of potassium from muscle cells into the bloodstream.
- Inform your doctor of all medications: diuretics, NSAIDs, ACE inhibitors, glucocorticoids, and lithium all affect electrolyte balance.
- The tourniquet should not compress the vein for more than one minute: prolonged stasis causes a false rise in potassium as ions leak from cells — pseudo-hyperkalaemia.
- For treatment monitoring, always test at the same laboratory at the same time of day.
A key technical note: haemolysis of the sample — breakdown of red blood cells during transport or storage — dramatically elevates potassium in the tube. If potassium is abnormally high with no symptoms, sample haemolysis is the first thing to exclude — not a pathological condition.
What the Panel Includes: Basic and Extended Versions
The composition of the panel depends on the clinical question. There are two standard formats.
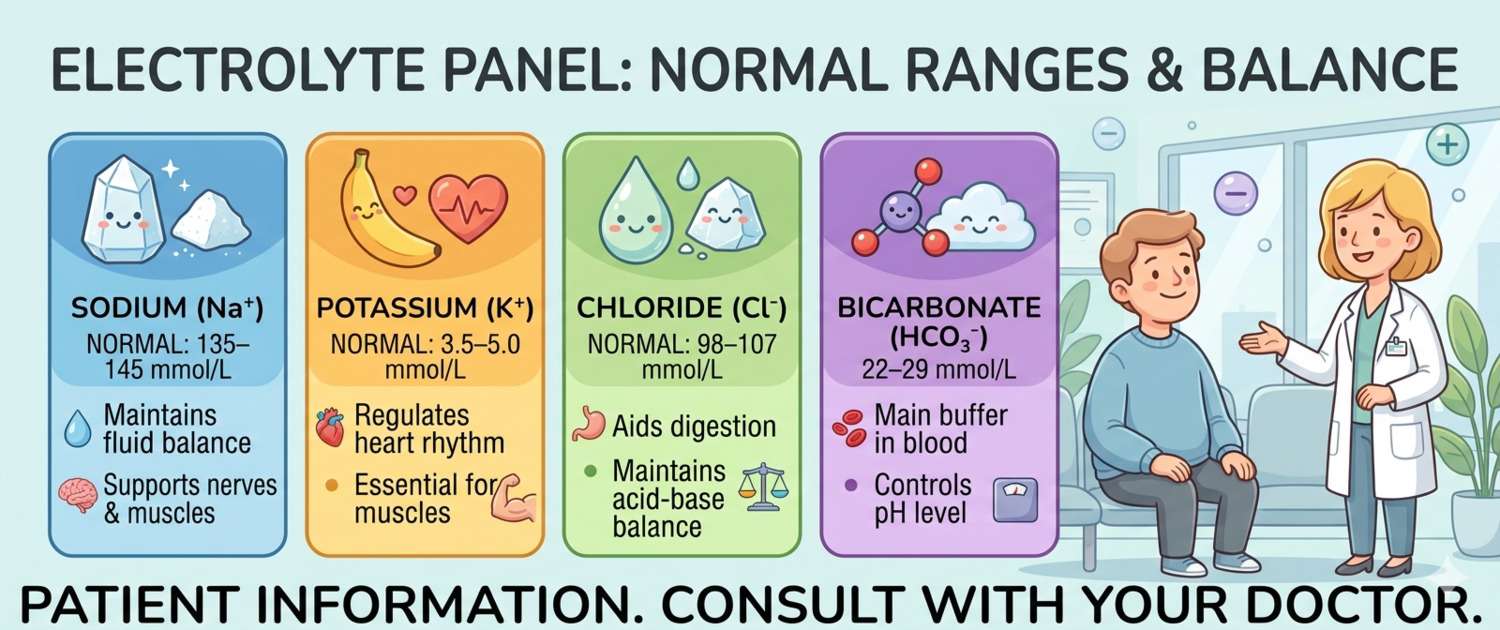
The basic metabolic panel includes four core electrolytes:
Sodium (Na⁺) — the main extracellular cation, determining plasma osmolality and circulating fluid volume. Regulated by aldosterone and antidiuretic hormone. Normal: 136–145 mmol/L. Sodium is the best marker of the body's fluid balance: its changes reflect not so much the sodium content itself as the ratio of water to salt in the body.
Potassium (K⁺) — the main intracellular cation. Although only 2% of total body potassium circulates in plasma, it is its plasma concentration that governs the excitability of muscles and the cardiac myocardium. Normal: 3.5–5.1 mmol/L. This is the most clinically critical electrolyte: a deviation of 1–2 mmol/L in either direction creates a real risk of life-threatening arrhythmia.
Chloride (Cl⁻) — the primary extracellular anion, following sodium to maintain electroneutrality. Normal: 98–107 mmol/L. Chloride is rarely assessed in isolation: its changes almost always mirror those of sodium or bicarbonate. Hypochloraemia in vomiting and hyperchloraemia in diarrhoea are classic patterns.
Bicarbonate (HCO₃⁻) — the principal buffer of acid-base balance. Normal: 22–29 mmol/L. A low level indicates metabolic acidosis (diabetic ketoacidosis, renal failure, lactic acidosis); a high level points to metabolic alkalosis (prolonged vomiting, diuretic overuse).
The extended panel adds:
Calcium (Ca²⁺) — regulates neuromuscular conduction, blood clotting, and bone mineralisation. Normal total calcium: 2.15–2.55 mmol/L. The ionised (free) fraction is the biologically active form; its level depends on blood pH and albumin concentration.
Magnesium (Mg²⁺) — a cofactor for over 300 enzymes; influences cardiac function, the nervous system, and muscle contraction. It frequently falls alongside potassium — and without correcting magnesium, normalising potassium can be impossible. Normal: 0.7–1.0 mmol/L.
Phosphorus (PO₄³⁻) — involved in energy metabolism (ATP) and bone mineralisation. Regulated by parathyroid hormone together with calcium. Normal: 0.87–1.45 mmol/L. It is chronically elevated in chronic kidney disease, accelerating vascular calcification.
Electrolyte Normal Values: Summary Table
Reference ranges may vary slightly between laboratories — always check the values on your own report.
| Electrolyte | Adults | Pregnancy | Children 1–12 years |
|---|---|---|---|
| Sodium (mmol/L) | 136–145 | 133–143 | 136–145 |
| Potassium (mmol/L) | 3.5–5.1 | 3.3–5.0 | 3.4–4.7 |
| Chloride (mmol/L) | 98–107 | 97–109 | 98–106 |
| Bicarbonate (mmol/L) | 22–29 | 18–25 | 20–28 |
| Total calcium (mmol/L) | 2.15–2.55 | 2.10–2.50 | 2.20–2.70 |
| Magnesium (mmol/L) | 0.7–1.0 | 0.65–1.0 | 0.7–1.0 |
| Phosphorus (mmol/L) | 0.87–1.45 | 0.80–1.40 | 1.29–1.78 |
In pregnant women, bicarbonate is physiologically lower due to compensatory respiratory alkalosis: faster breathing exhales more CO₂ and shifts the acid-base balance. This is a normal adaptation, not a pathological finding. Sodium is also slightly lower in pregnancy due to the physiological dilution of blood by an expanded plasma volume.
Interpreting Deviations: Potassium and Sodium Disorders
Doctors read the electrolyte panel as a whole, evaluating the interplay of ions alongside the clinical picture.
Potassium disorders — the most clinically dangerous.
Hypokalaemia (potassium < 3.5 mmol/L): muscle weakness, cramps, constipation, palpitations. On ECG — flattening of the T wave and appearance of U waves. In severe hypokalaemia (< 2.5 mmol/L) — risk of ventricular arrhythmia. Causes: thiazide and loop diuretics, vomiting and diarrhoea, primary hyperaldosteronism, Cushing's syndrome, glucocorticoid use.
Hyperkalaemia (potassium > 5.5 mmol/L): muscle weakness, ascending paralysis, bradycardia. On ECG — tall peaked T waves, widening of the QRS complex. At potassium above 7.0 mmol/L — risk of cardiac arrest. Causes: chronic kidney disease (leading cause), ACE inhibitors, spironolactone, adrenal insufficiency, massive tissue breakdown (rhabdomyolysis, tumour lysis syndrome).
Sodium disorders — the most common in clinical practice.
Hyponatraemia (sodium < 136 mmol/L) — the most prevalent electrolyte disorder in hospitalised patients. Symptoms depend on the rate of onset: chronic reduction causes minimal symptoms; acute reduction causes nausea, headache, confusion, seizures, and cerebral oedema. Causes are varied: heart failure, liver cirrhosis, syndrome of inappropriate ADH secretion (SIADH), hypothyroidism, thiazide diuretics, psychogenic polydipsia.
Hypernatraemia (sodium > 145 mmol/L): thirst, dry mucous membranes, confusion; in severe cases — seizures and coma. It almost always reflects a water deficit relative to sodium: diabetes insipidus, significant fluid losses (fever, severe diarrhoea) without adequate replacement.
Calcium and magnesium disorders.
Hypocalcaemia manifests as tetany — painful muscle spasms, Chvostek's sign (facial muscle twitch on tapping the facial nerve), and Trousseau's sign. Hypercalcaemia — "bones, stones, and groans": osteoporosis, kidney stones, weakness, and depression. Hypomagnesaemia accompanies hypokalaemia and hypocalcaemia, rendering both refractory to treatment until magnesium is corrected simultaneously.
When Electrolyte Imbalances Are Life-Threatening
Most moderate electrolyte abnormalities can be managed with scheduled outpatient care. But the following situations are medical emergencies:
- Potassium above 6.5 mmol/L or below 2.5 mmol/L — regardless of symptoms
- Any potassium abnormality combined with ECG changes
- Sodium below 120 mmol/L, or an acute fall of more than 10 mmol/L within 24 hours
- Calcium above 3.5 mmol/L — hypercalcaemic crisis: nausea, vomiting, confusion, coma
- Bicarbonate below 10 mmol/L — severe metabolic acidosis
- Any electrolyte abnormality in a pregnant woman — immediate consultation
- Multiple concurrent electrolyte abnormalities — a sign of serious systemic disease
A dangerous pitfall in treating hyponatraemia is correcting it too quickly. Raising sodium faster than 8–10 mmol/L per day causes osmotic demyelination — irreversible damage to the nerve fibres of the brain. This is precisely why electrolyte correction must always be managed by a physician.
Conclusion
An electrolyte panel is a fast, accurate tool for assessing the ionic balance on which the heart, muscles, kidneys, and nervous system all depend. The four core ions — sodium, potassium, chloride, and bicarbonate — are read together as an interconnected system: a change in one almost always triggers compensatory shifts in the others. Proper preparation — eliminating factors that cause pseudo-abnormalities such as prolonged tourniquet use, haemolysis, or a potassium-rich meal beforehand — ensures a reliable result. If you experience cramps, palpitations, or significant weakness after vomiting or diarrhoea, do not delay testing and medical assessment.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
Electrolytes are charged mineral ions — sodium, potassium, calcium, magnesium, and others — dissolved in the blood. They regulate fluid balance, nerve impulses, and muscle contraction, including the heart. Testing is indicated whenever balance disruption is suspected — cramps, arrhythmia, or pronounced weakness after vomiting or diarrhoea — as well as routinely in kidney disease, diabetes, and long-term diuretic use.
Because the plasma potassium concentration directly determines the excitability of the cardiac muscle. Even a deviation of 1–2 mmol/L above or below normal can trigger life-threatening arrhythmias. Hyperkalaemia from chronic kidney disease and hypokalaemia from diuretics are two of the most common causes of emergency hospitalisation for cardiac rhythm disturbances.
Hyponatraemia — sodium below 136 mmol/L — is the most common electrolyte disorder in hospitalised patients. With a chronic, gradual onset, symptoms are minimal; with acute onset, it causes headache, nausea, confusion, and cerebral oedema. A particularly dangerous trap is correcting it too fast: raising sodium by more than 10 mmol/L per day causes osmotic demyelination — irreversible brain damage. Treatment must always be physician-supervised.
The kidneys are the primary regulators of electrolyte balance, filtering blood and reabsorbing the precise amounts of each ion needed. As kidney filtration declines, electrolytes fail to be excreted adequately: potassium and phosphorus accumulate, while sodium and bicarbonate may fall. This is why electrolytes are checked at every visit in chronic kidney disease. The link between phosphorus accumulation and kidney disease progression is explained in detail in the phosphorus indicator article.
The two ions are physiologically linked: magnesium is required for the function of the potassium pumps in the cell membrane, which keep potassium inside the cell. When magnesium is deficient, these pumps malfunction and potassium leaks out into the urine. This is why, in refractory hypokalaemia that does not respond to potassium replacement, a doctor will always check magnesium — correcting it is a prerequisite for successfully raising potassium.
In some situations, yes — urine electrolyte measurement is needed to identify the cause of a blood abnormality. For example, in hyponatraemia, urinary sodium concentration helps distinguish renal from non-renal losses. In hypokalaemia, urinary potassium excretion reveals whether the loss is through the kidneys or the gastrointestinal tract. This is done through a urinalysis with electrolyte measurement, ordered in addition to the main blood panel.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service