Calcium in Blood (Ca): Normal Levels, Causes of Abnormalities
Every time your heart contracts, calcium is what triggers the muscle cells to act. When a nerve fires, when a blood clot forms, when a bone bears load — calcium is involved in all of it. A calcium blood test is part of standard biochemistry and can reveal parathyroid disorders, kidney disease, or nutritional deficiencies long before symptoms appear. This article covers what is measured, what the norms are, and what each type of deviation means.
What Is Calcium in Blood and Why Is It Measured
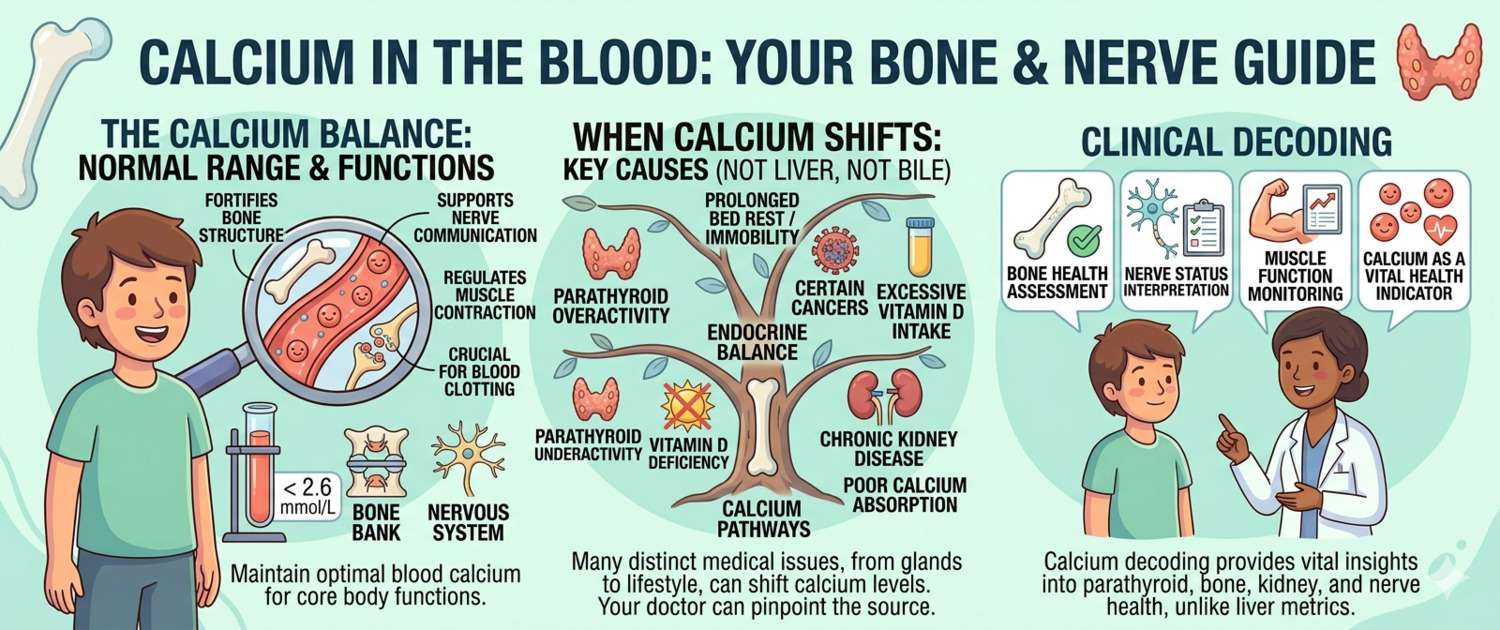
The human body contains about 1–1.5 kg of calcium. Roughly 99% is stored in bones and teeth as a strategic reserve. Only 1% circulates in the blood and tissues — but this fraction is critical for every cell in the body.
Calcium exists in three forms in serum:
- Ionized (free) calcium — about 50% of the total. The biologically active form that directly regulates muscle contraction, nerve conduction, hormone secretion, and blood clotting.
- Protein-bound calcium — 40–45%, held mainly by albumin. Biologically inactive, it acts as a reservoir for maintaining the ionized fraction.
- Complexed calcium — 5–10%, bound to citrate, phosphate, and bicarbonate. Also inactive.
A standard biochemical panel measures total calcium — the sum of all three forms. Ionized calcium can be ordered separately and more accurately reflects physiological activity. This distinction matters: when albumin is low, total calcium appears falsely reduced even when the ionized fraction is normal, which is why doctors use the corrected calcium formula.
Normal Calcium Levels: Total and Ionized
Reference ranges depend on age and the specific laboratory method.
Total Calcium Norms
| Age | Normal range, mmol/L |
|---|---|
| Newborns (up to 10 days) | 1.90–2.60 |
| Children under 2 years | 2.25–2.75 |
| Children 2–14 years | 2.20–2.70 |
| Adults 18–60 years | 2.15–2.55 |
| Adults over 60 years | 2.10–2.50 |
Differences of ±0.05 mmol/L between laboratories are normal. Always use the reference range printed on your own lab report.
Ionized Calcium Norms
| Group | Normal range, mmol/L |
|---|---|
| Adults | 1.15–1.30 |
| Children | 1.22–1.40 |
Corrected Calcium When Albumin Is Low
If albumin is below 40 g/L, apply the correction formula:
Corrected Ca (mmol/L) = measured Ca + 0.02 × (40 − albumin, g/L)
For example, with albumin at 28 g/L and total calcium at 2.05 mmol/L, the corrected value is 2.05 + 0.02 × 12 = 2.29 mmol/L — within normal range. Without this calculation, the patient would receive a false diagnosis of hypocalcemia.
How to Properly Prepare for a Calcium Blood Test
A few points that genuinely affect the result and are rarely explained.
Fasting. Blood should be drawn fasting — last meal 8–12 hours before. Avoid dairy products, nuts, and calcium supplements the evening before.
Calcium and vitamin D supplements. If you take supplements, inform your doctor: they may temporarily elevate the result. Some are paused 48–72 hours before testing; others are not — this is a clinical decision.
Tourniquet time. Prolonged tourniquet application (over 1 minute) before venipuncture can raise total calcium by 5–10% due to local hemoconcentration. Quality labs follow proper protocols.
Exercise. Intense physical activity has a minor effect on ionized calcium — avoid vigorous exercise 24 hours before the test.
Repeat testing. Any abnormal result should be repeated in the same laboratory before conclusions are drawn. A single reading is not a diagnosis.
High Calcium in Blood: Causes of Hypercalcemia
Hypercalcemia is total calcium above 2.55–2.60 mmol/L. Mild elevation (up to 3.0 mmol/L) is often asymptomatic and found incidentally. Above 3.0 mmol/L: fatigue, nausea, constipation, excessive urination. Above 3.5 mmol/L — a medical emergency: confusion, arrhythmias, risk of acute kidney injury.
| Cause | Mechanism |
|---|---|
| Primary hyperparathyroidism | Adenoma or hyperplasia of parathyroid glands — excess PTH mobilizes Ca from bones |
| Malignancy | Bone metastases or tumor-secreted PTHrP (parathyroid hormone-related protein) |
| Vitamin D toxicity | Excess calcitriol increases Ca absorption in the gut |
| Sarcoidosis and granulomatoses | Activated macrophages produce calcitriol independently |
| Thiazide diuretics | Reduce urinary calcium excretion |
| Prolonged immobilization | Accelerated bone resorption elevates Ca in blood |
| Milk-alkali syndrome | Excess combined intake of calcium and alkaline antacids |
| Hyperthyroidism | Accelerated bone turnover |
Primary hyperparathyroidism and malignancy together account for over 90% of hypercalcemia cases in outpatient adults. When elevated calcium is found, these two conditions are ruled out first.
Low Calcium in Blood: Causes of Hypocalcemia
Hypocalcemia is total calcium below 2.10–2.15 mmol/L or ionized calcium below 1.15 mmol/L. Symptoms range from tingling in the fingertips and around the mouth to severe muscle cramps. Severe hypocalcemia can cause laryngospasm and life-threatening cardiac arrhythmias.
| Cause | Mechanism |
|---|---|
| Hypoparathyroidism | PTH deficiency — Ca not mobilized from bone, renal reabsorption reduced |
| Vitamin D deficiency | Impaired Ca absorption in the small intestine |
| Chronic kidney disease | Impaired vitamin D activation, phosphate retention — double blow to calcium metabolism |
| Hypomagnesemia | Blocks PTH secretion and reduces tissue sensitivity to it |
| Acute pancreatitis | Calcium binds to fatty acids in the zone of fat necrosis |
| Hypoalbuminemia | Reduced protein-bound fraction (ionized calcium usually normal) |
| Hungry bone syndrome | Post-parathyroidectomy — bones rapidly absorb Ca from blood |
| Massive blood transfusions | Citrate preservative chelates ionized calcium |
| Celiac disease / malabsorption | Impaired absorption of Ca and vitamin D in the intestine |
Before diagnosing true hypocalcemia, the clinician always checks albumin and applies the correction formula if needed — to rule out a spuriously low result caused by low protein.
Calcium, PTH, and Vitamin D: The Regulatory Triangle
Calcium levels do not exist in isolation — they are maintained by three interlinked regulators.
Parathyroid hormone (PTH) is released by the parathyroid glands within seconds of a calcium drop. It acts on three fronts simultaneously: it mobilizes Ca from bone, increases Ca reabsorption in the kidneys, and stimulates production of active vitamin D to enhance intestinal Ca absorption.
Vitamin D (calcitriol) is activated in the kidneys under PTH control. Its main role in calcium metabolism is to drive Ca absorption in the small intestine. When vitamin D is deficient, the body cannot properly absorb calcium from food — regardless of how much is consumed.
Phosphorus is calcium's functional antagonist. Rising phosphate stimulates PTH secretion and simultaneously suppresses renal vitamin D activation. This is the mechanism behind secondary hyperparathyroidism in chronic kidney disease with phosphate retention — elevated PTH alongside low calcium.
This triangle explains clinical practice: when calcium is abnormal, doctors typically order a panel — PTH, vitamin D, phosphorus, and magnesium. Calcium alone tells only half the story.
When to See a Doctor Urgently
Many calcium disorders progress silently and require planned investigation. But certain signs call for immediate action.
Signs suggesting hypercalcemia:
- Confusion, drowsiness, or progressive weakness
- Persistent vomiting, abdominal pain, or intense thirst
- Irregular heartbeat
- Calcium above 3.0 mmol/L on a lab result — even without symptoms
Signs suggesting hypocalcemia:
- Muscle cramps or convulsions that do not resolve spontaneously
- Stridor (wheezing, labored breathing) — a sign of laryngospasm
- Numbness and tingling spreading to the face and trunk
- Ionized calcium below 0.90 mmol/L on a lab result
If your test shows an abnormality for the first time — do not panic, but do not delay. Repeat the test and see your GP: they will review the full picture and if necessary order further tests such as PTH, vitamin D levels, or a parathyroid ultrasound. Taking calcium supplements on your own without understanding the cause is unsafe — too much calcium is just as dangerous as too little and can lead to kidney stones, vascular calcification, and arrhythmias.
This article is for informational purposes only. Interpretation of test results and diagnosis are the responsibility of a qualified physician.
Frequently Asked Questions
Normal total calcium for adults aged 18–60 is 2.15–2.55 mmol/L (8.6–10.2 mg/dL). For ionized calcium, the normal range is 1.15–1.30 mmol/L. After age 60, total calcium reference values decrease slightly to 2.10–2.50 mmol/L. Always use the reference range listed on your own lab report, as values vary between analyzers and reagents.
The most common causes of hypercalcemia are primary hyperparathyroidism (adenoma or hyperplasia of the parathyroid glands) and malignancy with bone involvement. Together they explain over 90% of cases in outpatient adults. Less common causes include vitamin D toxicity, sarcoidosis, thiazide diuretics, and prolonged immobilization. Mild hypercalcemia (up to 3.0 mmol/L) often has no symptoms and is found incidentally on routine panels.
Hypocalcemia (total Ca below 2.10 mmol/L) most commonly results from vitamin D deficiency, hypoparathyroidism, or chronic kidney disease. Symptoms include tingling in the hands and around the mouth, muscle spasms, and in severe cases convulsions. An important caveat: low total calcium with low albumin may not represent true hypocalcemia — the ionized fraction is often normal. More on how kidneys affect calcium balance in the article on chronic kidney disease.
Total calcium is the sum of all three fractions: free ionized Ca, protein-bound Ca (mainly albumin), and complexed Ca. Ionized calcium is only the biologically active free fraction that directly regulates cellular functions. When protein levels are normal, both tests correlate well. When albumin is low — such as in liver disease or malnutrition — total calcium falls while ionized calcium may remain normal. In these situations, ionized calcium is more clinically informative.
Often yes. Calcium, sodium, potassium, magnesium, and phosphorus are closely interrelated. Low magnesium, for example, blocks PTH secretion and makes hypocalcemia resistant to treatment until magnesium is corrected. When mineral balance disorders are suspected, doctors typically order an electrolyte panel, which allows assessment of the full mineral picture in a single draw.
It is not advisable. Taking calcium supplements without a prior blood test and medical guidance carries real risks. Excess calcium — hypercalcemia — leads to kidney stones, vascular calcification, and cardiac arrhythmias. Before supplementing, you need to confirm that calcium is actually low and identify the underlying cause. For example, if the root cause is vitamin D deficiency, calcium supplements without vitamin D are largely ineffective since the intestine cannot absorb calcium without it.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service