Potassium in Blood: Normal Levels, Causes and Treatment
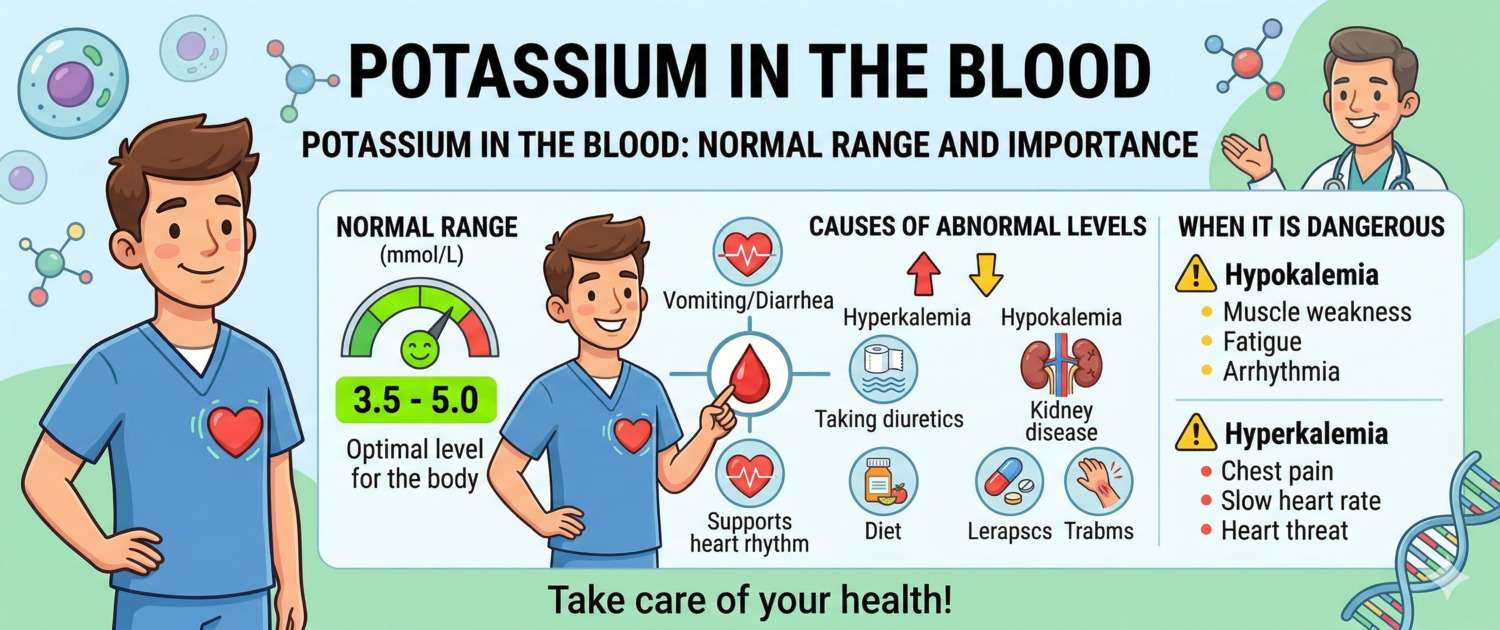
Potassium is one of the few electrolytes where a deviation in either direction can stop the heart. This is not an exaggeration: both hyperkalemia and severe hypokalemia are life-threatening — and both frequently develop without any warning symptoms until a critical threshold is crossed. This is precisely why potassium is a mandatory component of the electrolyte panel at most hospitalizations and of routine monitoring in patients with kidney disease, heart failure, or on diuretic therapy.
What Potassium Does and Why the Body Needs It
Potassium is the dominant intracellular cation: approximately 98% of the body's total potassium is inside cells, and only 2% in extracellular fluid, including blood plasma. This ratio — roughly 40:1 between intracellular and extracellular potassium — establishes the resting membrane potential, without which no electrical event in the body is possible.
Core functions of potassium:
- Action potential generation in nerve and muscle cells — including cardiomyocytes. Transmembrane potassium flow drives repolarization after every heartbeat
- Intracellular osmotic balance — maintained jointly with extracellular sodium
- Blood pressure regulation — through effects on vascular tone and renal sodium excretion: adequate dietary potassium intake measurably lowers blood pressure
- Glucose metabolism — insulin drives potassium from plasma into cells; this mechanism is exploited therapeutically in treating hyperkalemia
- Acid-base balance — potassium-hydrogen exchange participates in maintaining blood pH
Potassium is regulated primarily by the kidneys, which excrete or reabsorb it according to plasma levels under aldosterone control. Adrenal cortisol in excess exerts a mineralocorticoid effect — retaining sodium and expelling potassium — which explains hypokalemia in Cushing's syndrome.
Normal Blood Potassium Levels by Age
Plasma potassium is maintained within a narrow range. Even small deviations carry clinical significance.
| Age | Normal potassium (mmol/L) |
|---|---|
| Newborns 0–7 days | 3.7–6.0 |
| Infants 1 week – 6 months | 4.1–6.7 |
| Infants 6 months – 1 year | 3.7–6.0 |
| Children 1–2 years | 4.0–5.6 |
| Children 2–14 years | 3.4–4.7 |
| Adults | 3.5–5.1 |
| Elderly over 65 years | 3.5–5.3 |
In newborns and young infants, the normal range is higher than in adults — reflecting physiologically higher intracellular potassium content in growing tissues.
Clinically relevant thresholds in adults:
| K⁺ level (mmol/L) | Condition |
|---|---|
| > 6.5 | Severe hyperkalemia — high arrhythmia risk |
| 5.5–6.5 | Moderate hyperkalemia |
| 5.1–5.5 | Mild hyperkalemia |
| 3.5–5.1 | Normal |
| 3.0–3.5 | Mild hypokalemia |
| 2.5–3.0 | Moderate hypokalemia |
| < 2.5 | Severe hypokalemia — high arrhythmia risk |
How to Prepare for a Potassium Blood Test
Potassium is one of the most pre-analytically sensitive electrolytes. Incorrect collection or handling can produce falsely elevated results — pseudohyperkalemia — which is one of the most common sources of electrolyte misdiagnosis.
Pseudohyperkalemia occurs with:
- Sample hemolysis — red cell rupture from vigorous tube shaking, prolonged storage, or improper centrifugation releases intracellular potassium into plasma
- Prolonged tourniquet application during venipuncture — local muscle ischemia causes a localized potassium release
- Thrombocytosis or marked leukocytosis — clotting in the tube releases potassium from platelets and white cells (relevant when serum rather than plasma is used)
Collection rules:
- Draw fasting or 3–4 hours after a light meal
- Do not clench the fist during venipuncture — muscular work elevates local potassium
- Deliver the sample to the lab within 30–60 minutes
- When pseudohyperkalemia is suspected: repeat with a plasma sample (heparin tube, not serum) and centrifuge immediately
Inform the physician about potassium-sparing diuretics, ACE inhibitors, ARBs, and NSAIDs — all of these affect renal potassium excretion.
Causes of High Potassium (Hyperkalemia)
True hyperkalemia (after excluding pseudohyperkalemia) is a serious condition requiring immediate identification of the cause.
| Cause | Mechanism | Characteristic features |
|---|---|---|
| Chronic kidney disease | Reduced renal K⁺ excretion | Parallel rise in creatinine |
| Acute kidney injury | Sudden excretion failure | Oliguria, rising urea |
| Aldosterone deficiency (Addison's disease) | Absent excretion stimulus | Hyperkalemia + hyponatremia |
| Metabolic acidosis | K⁺ shifts out of cells in exchange for H⁺ | Low blood pH |
| Rhabdomyolysis, hemolysis | Massive K⁺ release from destroyed cells | Elevated CK, LDH |
| Tumor lysis syndrome | Tumor cell breakdown | During chemotherapy |
| Hyperkalemia-inducing drugs | Reduced excretion or cellular release | ACE inhibitors, ARBs, spironolactone, NSAIDs, trimethoprim |
| Excess K⁺ administration | Potassium supplement overdose | Iatrogenic |
| Decompensated diabetes | Insulin deficiency → K⁺ cannot enter cells | Hyperglycemia, ketoacidosis |
The most dangerous combination in clinical practice: hyperkalemia in a patient with chronic kidney disease who is taking an ACE inhibitor or ARB. Both drug classes reduce aldosterone secretion; the kidneys are already struggling to excrete potassium — and levels rise silently to a critical threshold.
Causes of Low Potassium (Hypokalemia)
Hypokalemia is far more common than hyperkalemia in ambulatory practice. The main routes of loss: renal, gastrointestinal, or intracellular redistribution.
| Cause | Mechanism | Characteristic features |
|---|---|---|
| Diuretics (thiazide, loop) | Enhanced renal K⁺ excretion | Most common clinical cause |
| Vomiting and diarrhea | GI K⁺ losses | With chronic disturbances |
| Primary hyperaldosteronism | Excess aldosterone → K⁺ excretion | Hypertension + hypokalemia |
| Cushing's syndrome | Mineralocorticoid effect of cortisol | Hypokalemia + hypertension + obesity |
| Metabolic alkalosis | K⁺ shifts into cells in exchange for H⁺ | Vomiting, antacids |
| Insulin / glucose administration | K⁺ driven into cells | During ketoacidosis treatment |
| Dietary deficiency | Insufficient K⁺ intake | Anorexia, extreme diets |
| Beta-2 agonists (salbutamol) | Na⁺/K⁺-ATPase stimulation | At high doses during bronchospasm treatment |
| Alcohol use disorder | Poor intake + urinary losses | Combined with hypomagnesemia |
| Hypomagnesemia | Mg²⁺ required to retain K⁺ inside cells | Hypokalemia refractory to treatment |
The last entry deserves special clinical emphasis: hypokalemia coexisting with hypomagnesemia cannot be corrected without simultaneously replenishing magnesium. The Mg²⁺ ion is required for Na⁺/K⁺-ATPase function — the pump that keeps potassium inside cells. Without magnesium, administered potassium immediately leaks back out. This is why persistent hypokalemia unresponsive to potassium supplementation should always prompt magnesium measurement.
Potassium and the Heart: ECG Changes in Dyscalemia
The heart is the primary target organ for potassium disturbances. Cardiomyocytes are acutely sensitive to the transmembrane potassium gradient because it drives the repolarization phase of every cardiac cycle.
ECG patterns in hyperkalemia — escalating with rising potassium:
- 5.5–6.5 mmol/L: tall, peaked T waves ("tented" T waves)
- 6.5–7.5 mmol/L: QRS widening, reduced P wave amplitude
7.5 mmol/L: sinusoidal ("sine wave") pattern, ventricular fibrillation, asystole
ECG patterns in hypokalemia:
- T wave flattening and inversion, prominent U wave (following T)
- QT interval prolongation — risk of polymorphic ventricular tachycardia (torsades de pointes)
- In severe hypokalemia — ventricular arrhythmias, particularly dangerous in patients with ischemic heart disease
Important: ECG changes in hyperkalemia may appear at levels above 5.5 mmol/L, and at levels above 7.0 mmol/L the risk of fatal arrhythmia is high even before symptoms develop. A normal ECG does not rule out dangerous hyperkalemia.
Hypokalemia potentiates cardiac glycoside toxicity (digoxin): at low potassium, digoxin binds to its receptor (Na⁺/K⁺-ATPase) with substantially greater affinity — dramatically increasing the risk of digitalis-induced arrhythmia. Potassium monitoring in patients on digoxin is non-negotiable.
When Potassium Abnormalities Require Medical Attention
Any potassium value outside the reference range warrants medical evaluation. The urgency is determined by the absolute level, symptoms, and comorbidities.
Scheduled visit to a doctor when:
- Potassium 5.1–5.5 mmol/L (mild hyperkalemia) — identify cause, especially in patients on ACE inhibitors/ARBs or with kidney disease
- Potassium 3.0–3.5 mmol/L (mild hypokalemia) without symptoms — review diuretic therapy and dietary intake
See a doctor within hours when:
- Potassium > 5.5 or < 3.0 mmol/L in any condition
- Muscle weakness, cramps, or paresthesias alongside any potassium deviation
Call emergency services immediately when:
- Potassium > 6.5 mmol/L — regardless of symptoms
- Potassium < 2.5 mmol/L — regardless of symptoms
- Cardiac arrhythmia (palpitations, irregular heartbeat, syncope) in the context of a known potassium disturbance
- Paralysis or progressive muscle weakness approaching respiratory compromise
Self-correcting potassium — taking supplements or restricting dietary potassium without laboratory monitoring — is genuinely dangerous. The rate of correction in severe hypokalemia is strictly controlled: rapid intravenous potassium administration without continuous ECG monitoring is potentially fatal.
This article is for informational purposes only and does not replace professional medical advice. Consult a GP or nephrologist if your blood potassium is outside the normal range.
Frequently Asked Questions
This phenomenon is called pseudohyperkalemia — one of the most common sources of incorrect electrolyte diagnosis. The mechanism: red blood cells contain 25–30 times more potassium than plasma. When the sample hemolyzes (from tube agitation, prolonged storage, or improper centrifugation), this intracellular potassium floods the plasma, significantly overstating the result. A similar effect comes from prolonged tourniquet application during venipuncture. When pseudohyperkalemia is suspected, the test is repeated immediately with correct technique — results frequently normalize.
Hypokalemia manifests primarily through muscle symptoms: weakness (especially in the legs), cramps, and muscle spasms. Constipation and bloating are common at moderate deficiency — intestinal motility slows. In severe hypokalemia, paralysis, cardiac arrhythmias, and respiratory failure are possible. Polyuria and thirst are characteristic when hypokalemia is of renal origin (e.g., hyperaldosteronism). Assessing kidney function in hypokalemia is best done with a kidney function test to identify a renal cause.
The kidneys are the primary route for potassium excretion — roughly 90% of the daily potassium load is cleared renally. When kidney function declines, the glomerular filtration rate falls and the kidneys lose the capacity to excrete the daily potassium load. The result is gradual accumulation in the blood. Hyperkalemia is one of the most dangerous complications of both chronic and acute kidney failure, requiring careful dietary restriction and close monitoring.
With normal kidney function, dietary potassium is almost entirely excreted and blood levels remain stable. However, in patients with impaired renal excretion, potassium-rich foods (bananas, potatoes, tomatoes, avocado, legumes, nuts, dried fruits) can significantly elevate blood levels. In hypokalemia, conversely, a diet enriched with these foods is an important adjunct to medical correction. Calcium in blood often falls alongside potassium in conditions that drive losses of both electrolytes simultaneously.
Yes — this risk is consistently underestimated. Potassium in supplements and vitamins is often perceived as a harmless 'natural' additive, but in patients with impaired renal excretion or those taking ACE inhibitors or ARBs, additional potassium intake can cause severe hyperkalemia with fatal arrhythmias. Self-supplementing potassium is only reasonable when a confirmed deficiency exists on laboratory testing, kidney function is normal, and none of the above-mentioned drug interactions are present.
The most common cause of refractory hypokalemia is co-existing magnesium deficiency. The Mg²⁺ ion is required for Na⁺/K⁺-ATPase — the pump that retains potassium inside cells. When magnesium is depleted, the pump functions poorly: administered potassium immediately leaks back out of cells and is excreted by the kidneys. Without magnesium repletion, normalizing potassium is impossible. Chronic heavy alcohol use is the classic cause of simultaneous depletion of both electrolytes.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service