Acute Pancreatitis: Symptoms, Causes, Diagnosis and Treatment
The pain of acute pancreatitis is described with striking consistency — a band-like, unbearable sensation that allows no comfortable position. This is not coincidence: the inflamed pancreas literally begins digesting itself. Acute pancreatitis is one of the most common abdominal emergencies and requires immediate hospitalisation. Around 80% of cases are mild and resolve within days. The remaining 20% are severe — with pancreatic necrosis, multi-organ failure and significant mortality. Here is how the disease develops, how it is diagnosed, and what modern treatment looks like.
What Is Acute Pancreatitis and How Does It Develop
The pancreas serves two functions. Its exocrine component produces digestive enzymes — amylase, lipase, proteases — and releases them into the duodenum in inactive form (zymogens). Activation happens only there, in the intestine, under the action of enterokinase. This is a protective mechanism: if enzymes were activated inside the gland, it would digest itself.
In acute pancreatitis, exactly that happens. A trigger — ductal obstruction, toxic cell damage or direct trauma — causes enzymes to activate prematurely, inside the acinar cells themselves. An autolysis cascade is set off: proteases destroy cell membranes, phospholipase A2 attacks membrane phospholipids, elastase breaks down vessel walls. The inflammatory response escalates rapidly — involving adjacent tissues, retroperitoneal fat, and in severe cases the entire body through systemic cytokine release.
Critically: the severity of pancreatitis is determined not only by the extent of gland injury but by the systemic response — the development of organ dysfunction. This is why mild pancreatitis is managed in a medical ward and severe pancreatitis in the intensive care unit.
Symptoms of Acute Pancreatitis
The clinical picture of acute pancreatitis is characteristic enough that an experienced clinician often makes the provisional diagnosis in the emergency department.
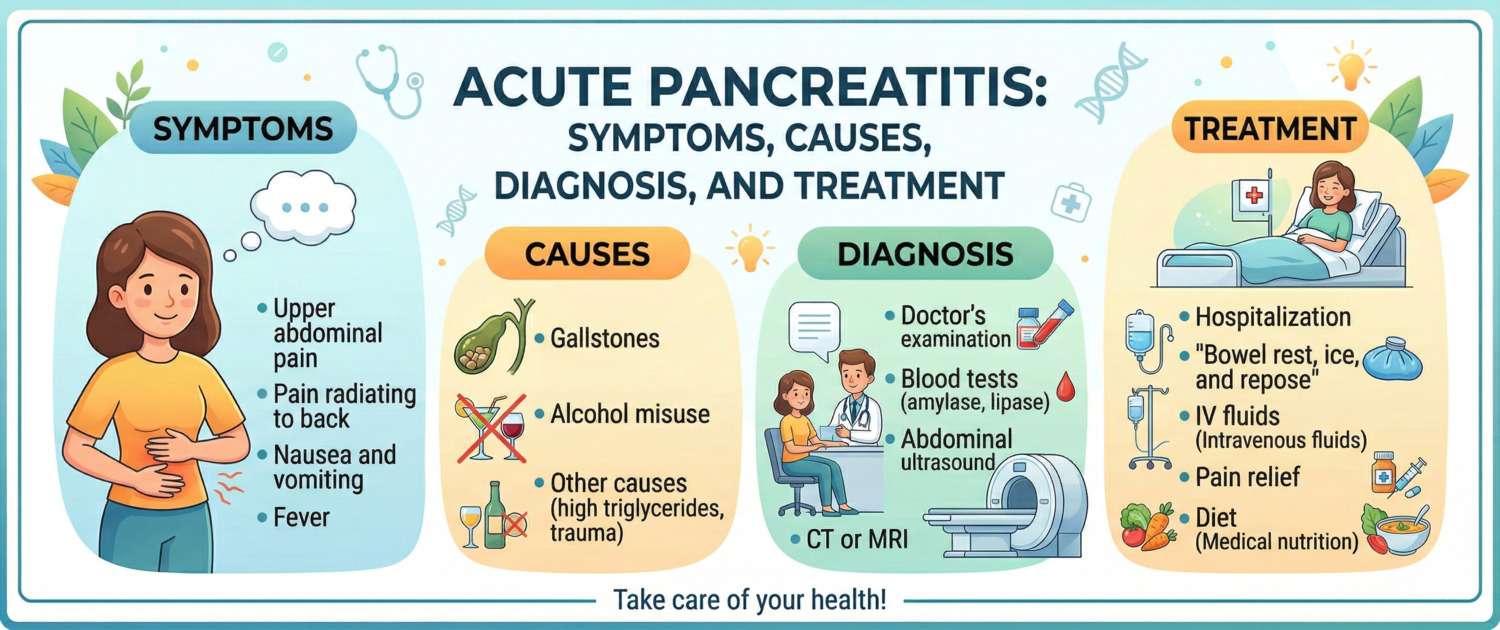
Pain — the central symptom. It begins suddenly, typically after a heavy fatty meal or alcohol. Location: epigastrium (upper abdomen), radiating to the back, left upper quadrant, or encircling the torso. The pain is intense and constant — unlike colic, it does not ease and does not change with breathing. The characteristic posture: knees drawn to the chest, lying on one side — slightly more bearable in this position.
Nausea and vomiting — almost always present. Vomiting is repeated and brings no relief — this is an important distinguishing feature. If pain eases after vomiting, pancreatitis is less likely.
Abdominal rigidity — mild in mild pancreatitis, pronounced in severe and necrotising forms. Peritoneal signs (board-like abdomen, rebound tenderness) appear when the process extends beyond the gland.
Systemic signs in severe forms:
- Tachycardia and falling blood pressure — shock from third-space fluid sequestration
- Fever above 38.5°C — sign of infected necrosis or systemic inflammation
- Jaundice — compression of the common bile duct by a swollen pancreatic head, or pre-existing choledocholithiasis
- Periumbilical bruising (Cullen's sign) or flank bruising (Grey-Turner's sign) — retroperitoneal haemorrhage; rare but ominous
Causes of Acute Pancreatitis
More than 80% of all acute pancreatitis cases are explained by two causes.
| Cause | Proportion | Mechanism |
|---|---|---|
| Gallstone disease (biliary pancreatitis) | 40–50% | Stone impaction at the ampulla of Vater → bile reflux into the pancreatic duct → enzyme activation |
| Alcohol | 25–35% | Direct acinar cell toxicity + Oddi sphincter spasm + altered pancreatic juice composition |
| Hypertriglyceridaemia | 1–4% | Triglycerides > 11 mmol/L → fat emboli in pancreatic vessels → ischaemia and inflammation |
| Hypercalcaemia | < 1% | Calcium activates trypsinogen within the gland |
| Trauma, ERCP | 1–3% | Direct mechanical duct injury |
| Medications | 1–2% | Azathioprine, furosemide, tetracyclines, valproic acid and others |
| Autoimmune pancreatitis | < 1% | IgG4-mediated inflammation |
| Idiopathic | 10–20% | No cause found after full investigation |
The link with metabolic syndrome is direct and underappreciated: obesity amplifies the severity of any pancreatitis regardless of cause, and hypertriglyceridaemia is an independent trigger. A patient with abdominal obesity and triglycerides above 10 mmol/L carries a high pancreatitis risk even without alcohol or gallstones.
Diagnosis: Blood Tests and Investigations
The diagnosis of acute pancreatitis is made when two of three Atlanta criteria are present:
- Characteristic abdominal pain
- Serum lipase or amylase ≥ 3× the upper limit of normal
- Characteristic findings on CT or MRI
In practice, most patients satisfy the first two criteria — CT is reserved for diagnostic uncertainty, severe presentation, or failure to improve within 48–72 hours.
Laboratory investigations:
Amylase and lipase levels are the key markers. Lipase is preferred: higher specificity (85–90%) and longer elevation. At ≥ 3× ULN combined with pain, the diagnosis is rarely in doubt.
C-reactive protein (CRP) — the primary severity marker. At 48 hours from onset, a CRP above 150 mg/L predicts severe pancreatitis with approximately 80% sensitivity. This timing window matters: in the first 24 hours CRP has not yet peaked, and a low value does not exclude severe disease.
Liver function tests — to assess biliary aetiology: ALT elevation above 3× ULN on admission is a sensitive indicator of gallstone pancreatitis. Also needed for baseline hepatic function assessment before treatment.
Full blood count: leucocytosis (> 12×10⁹/L) indicates inflammation; haematocrit > 44% signals haemoconcentration from fluid sequestration — a marker of necrosis risk. Falling platelets in severe cases suggest DIC.
Calcium, glucose, urea — monitoring for organ complications: hypocalcaemia (calcium sequestered in fat necrosis foci), hyperglycaemia (insulin-secreting cell damage), rising urea (pre-renal azotaemia from dehydration).
Imaging:
- Abdominal ultrasound — first step; detects gallstones, ductal dilatation, free fluid. The pancreas itself is often obscured by bowel gas during active pancreatitis.
- Contrast-enhanced CT — gold standard for severity assessment. Identifies necrosis (non-enhancing areas), peripancreatic collections, abscesses. Optimal at 48–72 hours — before this, necrosis is not yet fully demarcated.
- MRI / MRCP — when ductal anatomy needs assessment, or when CT is contraindicated (pregnancy, contrast allergy).
Severity scoring using the Balthazar CT index and systemic scales (APACHE II, BISAP) guides decision-making about care intensity in severe presentations.
Treatment of Acute Pancreatitis
Pancreatitis management has undergone a revolution over the past 20 years: from "nothing by mouth, nothing by vein" to aggressive fluid resuscitation and early nutritional support.
Mild and Moderate Pancreatitis
Fluid resuscitation — the foundation of treatment. Aggressive intravenous crystalloid administration (Ringer's lactate is preferred over normal saline — reduces acidosis risk) in the first 24–48 hours prevents pancreatic necrosis by maintaining perfusion. Target: urine output 0.5–1 mL/kg/hour.
Pain management — a priority from the first minutes. NSAIDs, ketamine, opioids — the choice depends on severity. The old fear that morphine causes Oddi sphincter spasm and worsens pancreatitis is not supported clinically — analgesia is not delayed.
Nutrition — a paradigm shift. Previously, patients were kept nil by mouth for 5–7 days. Today, in mild pancreatitis, oral feeding is recommended as early as 24–48 hours after pain subsides, starting with liquids. Fasting does not accelerate recovery and worsens gut dysfunction.
Antibiotics — only for confirmed infected necrosis (positive culture or CT showing gas within necrosis). Prophylactic antibiotics for sterile necrosis are not recommended — this is a well-established evidence base.
Severe Pancreatitis and Necrosis
- Intensive care — for multi-organ failure; monitoring of haemodynamics, renal function, coagulation
- Enteral nutrition via nasojejunal tube — preferred over parenteral; preserves the gut barrier, reduces bacterial translocation
- Endoscopic drainage (endoscopic necrosectomy under EUS guidance) — for infected necrosis; preferred over open surgery in most situations
- ERCP — for biliary pancreatitis with cholangitis or persistent bile duct obstruction; performed within the first 24–72 hours
- Cholecystectomy — after resolution of acute biliary pancreatitis; ideally in the same admission or within 4–6 weeks
In chronic hepatitis with gallstone disease, management is coordinated with a hepatologist — underlying liver disease alters the pharmacokinetics of most medications.
Complications of Acute Pancreatitis
Complications are divided into early (first 1–2 weeks) and late (after 2–4 weeks).
Early:
- Systemic inflammatory response syndrome (SIRS) — escalating cytokine storm; risk of progression to sepsis
- Multi-organ failure — renal (acute kidney injury), respiratory (ARDS), cardiovascular. This is what drives mortality in severe pancreatitis
- Acute peripancreatic fluid collections — fluid around the gland; most resolve spontaneously
- Hypocalcaemia — calcium is "captured" by fat necrosis foci; severe hypocalcaemia is an adverse prognostic sign
Late:
- Pseudocyst — an encapsulated collection of pancreatic juice forming 4–6 weeks after the attack; treatment needed only when symptomatic or infected
- Walled-off necrosis (WON) — mature encapsulated necrosis zone; requires drainage when infected
- Pancreatic duct fistulae — leak of ductal contents into the peritoneal or pleural cavity
- Exocrine and endocrine insufficiency — after extensive necrosis; the patient may require lifelong enzyme supplementation and possibly insulin therapy
When to See a Doctor Urgently
Acute pancreatitis always warrants hospitalisation. Call emergency services immediately for:
- Sudden, severe epigastric or band-like abdominal pain that does not ease — call an ambulance immediately
- Repeated vomiting without relief combined with abdominal pain
- Falling blood pressure, rapid pulse, pallor — signs of shock
- Jaundice with abdominal pain — probable biliary pancreatitis with bile duct obstruction
- Fever above 38.5°C with abdominal pain — possible infected necrosis
Do not wait for it to "pass by itself," and do not take antispasmodics or analgesics "to see what happens" — modern clinicians work on the opposite principle. Adequate analgesia does not impair diagnosis, and delayed hospitalisation in severe pancreatitis directly increases the risk of necrosis and death.
This article is for informational purposes only. Diagnosis and treatment are the responsibility of a surgeon or gastroenterologist.
Frequently Asked Questions
The diagnosis is confirmed by lipase or amylase ≥ 3× the upper limit of normal combined with characteristic pain. Mandatory tests include a full blood count (leucocytosis, haematocrit), biochemistry (amylase, lipase, CRP, glucose, urea), and liver function tests to assess a biliary cause. Calcium is measured to monitor complications: hypocalcaemia develops in severe necrosis and is an adverse prognostic sign.
No. Acute pancreatitis is an indication for hospitalisation in all cases. Even a mild episode requires intravenous fluid therapy, adequate pain relief and monitoring of enzyme dynamics. Attempting to manage it at home is dangerous: mild pancreatitis can progress to severe, and signs of deterioration are easily missed without laboratory and imaging surveillance.
In the acute phase — nothing by mouth for 0–24 hours, intravenous fluids only. Once pain subsides, oral intake resumes with liquids: clear broth, rice water, diluted juices. Strictly avoided: fatty foods, fried food, alcohol, spicy food, carbonated drinks. Dietary restrictions continue for weeks to months after discharge depending on severity. The principle: minimal pancreatic stimulation — small frequent meals of low-fat boiled or steamed food.
Mild pancreatitis typically resolves within 5–7 days in hospital, after which most patients feel well. Moderate pancreatitis with peripancreatic collections may require 2–4 weeks of treatment. Severe necrotising pancreatitis involves months of treatment with possible repeat interventions and prolonged rehabilitation. In 10–30% of patients, a first episode progresses to chronic pancreatitis if the underlying trigger is not eliminated.
Yes, and this is common. In gallstone disease, a recurrent attack occurs in 30–50% of patients without cholecystectomy — which is precisely why surgery is recommended in the same admission or within 4–6 weeks. In alcoholic pancreatitis, recurrence risk depends entirely on cessation of alcohol. In hypertriglyceridaemia, it depends on controlling triglycerides. Each subsequent episode increases the probability of progression to chronic pancreatitis.
Severe pancreatitis with gland necrosis carries the risk of multi-organ failure: acute kidney injury, respiratory failure and septic shock when necrosis becomes infected. Mortality in severe pancreatitis is 15–30%; in infected necrosis without treatment it is higher. Even after successful management of extensive necrosis, the patient may be left with permanent exocrine insufficiency (impaired fat digestion) and diabetes. More on kidney involvement in systemic illness in the chronic kidney disease article.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service