Endometriosis: Symptoms, Diagnosis and Treatment Options
One in five women with endometriosis is told by a doctor that her pain is just "normal period pain" — and sent home with painkillers. On average, seven to ten years pass between the first symptoms and a confirmed diagnosis. During that time, endometriotic lesions grow, form adhesions and quietly erode fertility. Understanding what is happening in the body is the first step toward the right diagnosis.
What Endometriosis Is and How It Develops
Endometriosis is a chronic, hormone-dependent disease in which tissue resembling the endometrium — the inner lining of the uterus — is found and functions outside the uterine cavity. These lesions respond to cyclic oestrogen fluctuations: they swell in the luteal phase and "bleed" during menstruation — but the blood from ectopic deposits has nowhere to drain. It accumulates, triggers an inflammatory reaction, and leads to adhesions and "chocolate cysts" (endometriomas).
The most widely accepted theory of origin is retrograde menstruation: part of the menstrual blood carrying endometrial cells passes through the fallopian tubes into the peritoneal cavity. This occurs in most women, but a normally functioning immune system destroys these displaced cells. In endometriosis, something in this mechanism is defective: the cells implant, survive and proliferate. Genetic predisposition explains why first-degree female relatives of affected women face a seven- to tenfold higher risk.
The location of lesions determines the clinical picture. The most commonly affected sites are the ovaries (endometriomas), the rectouterine pouch, the uterosacral ligaments and the pelvic peritoneum. In deep infiltrating endometriosis (DIE), lesions penetrate the bladder, ureters, rectum and rectovaginal septum.
Staging by the rASRM classification (I–IV) reflects the extent and severity of adhesions but correlates poorly with pain. Clinically, it is more meaningful to distinguish between superficial peritoneal endometriosis, ovarian endometriomas and DIE — the last requiring a fundamentally different surgical approach.
Causes and Risk Factors
The precise cause of endometriosis is unknown. The contemporary model regards it as multifactorial — genetic susceptibility, immune dysregulation, hormonal imbalance and environmental factors all interact.
Genetics: a mother or sister with endometriosis increases risk seven- to tenfold. Several susceptibility loci have been identified but no single "endometriosis gene" exists.
Hormonal environment: endometriosis is oestrogen-dependent. Lesions contain excess aromatase enzyme, which synthesises oestradiol directly at the implantation site — local hyperoestrogenaemia sustains growth. Simultaneously, resistance to progesterone is impaired.
Immune dysfunction: peritoneal NK cells and macrophages are less effective at eliminating ectopic endometrial cells. Pro-inflammatory cytokines in the peritoneal fluid create conditions favourable for implantation and growth.
Factors that increase risk: early menarche (before 12), short cycles (< 27 days), heavy periods, nulliparity, low BMI.
Factors that reduce risk: pregnancies, lactation, long-term oral contraceptive use, regular physical exercise.
An important distinction: polycystic ovary syndrome and endometriosis are not the same condition, though both impair reproductive function. PCOS is characterised by androgen excess and anovulation; endometriosis by hyperoestrogenaemia and pain. In some women both coexist, substantially complicating diagnosis.
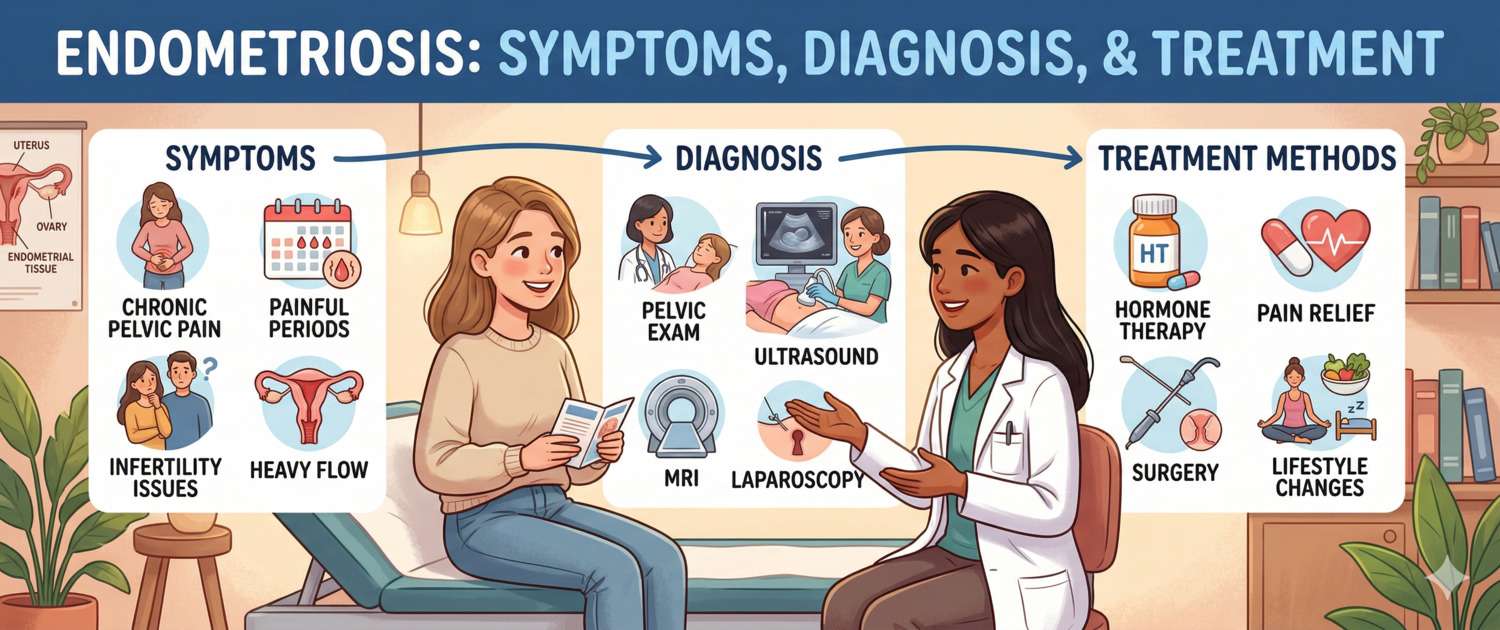
Symptoms: Pain and Infertility
The two hallmarks of endometriosis are chronic pelvic pain and reduced fertility. Their severity does not correlate with disease stage.
Pain:
- Dysmenorrhoea — painful periods not controlled by standard NSAIDs. Periods that are disabling for one to two days a month require investigation, not simply painkillers
- Chronic pelvic pain — dull or cramping pain in the lower abdomen or spine persisting outside menstruation for more than 6 months
- Dyspareunia — painful intercourse with deep penetration. Classically associated with endometriosis of the posterior Douglas pouch
- Dyschezia — painful defecation worsening during menstruation. Characteristic of rectovaginal endometriosis
- Dysuria — painful urination intensifying before menstruation
Menstrual disturbances: heavy periods (menorrhagia), intermenstrual spotting, premenstrual brown discharge.
Infertility: endometriosis is found in 25–50% of women presenting with infertility. Adhesions distort the fallopian tubes; endometriomas reduce ovarian reserve; the peritoneal inflammatory milieu is toxic to sperm and embryos; immune abnormalities interfere with implantation.
Diagnosis: Laparoscopy, Ultrasound and Blood Tests
Laparoscopy with biopsy and histological confirmation remains the gold standard. However, current guidelines support a clinical diagnosis when history and ultrasound findings are characteristic — treatment may be started without surgery.
Transvaginal ultrasound — the first-line investigation. Sensitivity for ovarian endometriomas is 90–95%: the characteristic appearance is a unilocular cyst with homogeneous ground-glass echogenicity. MRI of the pelvis is best for mapping DIE involving the bowel and urinary tract.
Laparoscopy with biopsy — mandatory for atypical presentations, suspected DIE, or failure of hormonal therapy.
Laboratory tests:
CA-125 — elevated in 75–80% of stage III–IV patients but only 20–25% at stage I–II. The main value is monitoring: a rising level after surgery or during treatment signals progression. CA-125 above 200 U/mL in an endometrioma requires extended oncological evaluation with HE4 and the ROMA index.
Sex hormone panel — FSH, LH, oestradiol, AMH. AMH is critical: endometriomas reduce ovarian reserve, and AMH below 1.0 ng/mL before surgery signals risk of further depletion. This determines the approach for women planning pregnancy.
Complete blood count — heavy periods frequently cause iron deficiency anaemia: recurrent large blood loss depletes iron stores progressively.
Endometriosis and the Risk of Ovarian Cancer
The link between endometriosis and ovarian cancer is real and clinically significant: women with endometriosis have approximately three times the population risk of ovarian cancer. But the absolute risk remains low — the vast majority of women with endometriosis never develop cancer.
The histological subtypes most closely associated with endometriosis are clear cell carcinoma and endometrioid carcinoma. The mechanism is malignant transformation of endometrioma cells driven by chronic oxidative stress and inflammation.
Features associated with elevated transformation risk: disease duration above 10 years, large endometriomas (> 9 cm), growth of the cyst or new solid component on follow-up imaging, age over 45, postmenopausal status. When these features are present, ultrasound intervals are shortened to 3 months and HE4 with ROMA index calculation is added.
Treatment: Hormones, Surgery and Pain Management
Treating endometriosis is a long-term strategy. Three factors determine the approach: severity of pain, desire to preserve fertility, and stage and location of lesions.
Hormonal therapy:
Combined oral contraceptives (COCs) — first-line for dysmenorrhoea and chronic pelvic pain. A continuous regimen (no placebo interval) is more effective than cyclic dosing.
Progestins (dienogest, medroxyprogesterone) — as effective as COCs for pain; dienogest is superior for deep endometriosis and reduces endometrioma size with prolonged use.
GnRH agonists (leuprorelin, buserelin) — induce medical menopause. Effective for severe pain and IVF preparation. Mandatory "add-back" low-dose oestrogen-progestin therapy protects bone density; without it, treatment is limited to 6 months.
Surgical treatment:
Laparoscopic lesion excision is preferred over coagulation: it provides histological confirmation and lower recurrence risk. For endometriomas: cystectomy reduces recurrence but irreversibly reduces ovarian reserve. AMH must be measured before ovarian surgery in women planning pregnancy.
DIE involving the bowel or urinary tract requires surgery in a specialist endometriosis centre with a gynaecologist, colorectal surgeon and urologist.
Fertility treatment: stage I–II with patent tubes — ovulation induction with or without IUI. Stage III–IV or previous treatment failure — IVF.
When to See a Doctor
Painful periods that interfere with daily life are not a normal variant and require investigation, not simply analgesics.
See a gynaecologist when one or more of the following apply:
- dysmenorrhoea severe enough to require prescription pain relief or to miss work or studies;
- chronic pelvic pain outside menstruation lasting more than 3 months;
- deep dyspareunia;
- difficulty conceiving after 6–12 months of regular unprotected intercourse;
- an ovarian cyst with endometrioma characteristics found incidentally;
- a mother or sister has been diagnosed with endometriosis.
Endometriosis is a chronic condition without a definitive cure, but effective symptom control and fertility preservation are achievable. Early diagnosis and a long-term treatment strategy agreed with a specialist are the keys to a better prognosis.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
Endometriosis-associated dysmenorrhoea characteristically progresses over time, becoming more severe and longer-lasting year by year. It is not controlled by standard doses of ibuprofen, is disabling for one to two days, and is typically accompanied by pain during intercourse or defecation. Primary dysmenorrhoea (without a structural cause) tends to ease with age or after childbirth. An accurate distinction requires gynaecological examination, transvaginal ultrasound and — when indicated — laparoscopy.
Yes. At stage I–II, most women conceive naturally, though it may take longer. At stage III–IV with significant adhesions or large endometriomas, spontaneous conception is less likely. Ovulation induction with IUI and IVF are effective options. Before ovarian surgery, AMH must be measured — removing an endometrioma reduces ovarian reserve and this must be factored into fertility planning. Chronic anaemia from heavy menstrual loss reduces endometrial receptivity: a falling haemoglobin is a practical reason to assess iron status before starting fertility treatment.
Adenomyosis is a condition in which endometrial tissue invades the myometrium, causing uterine thickening. Symptoms are similar — painful heavy periods, chronic pelvic pain — but the mechanism is different. Adenomyosis is diagnosed by MRI or ultrasound (characteristic honeycomb myometrial pattern). Both conditions can coexist in the same patient. Treatment is broadly analogous: progestins, GnRH agonists, the levonorgestrel intrauterine system, and hysterectomy in severe cases.
In absolute terms, rarely: the risk of malignant transformation of an endometrioma is approximately 0.5–1% over the entire follow-up period. However, the relative risk of ovarian cancer in women with endometriosis is three times higher than in the general population. Warning signs requiring urgent oncological review: rapid growth of the endometrioma, appearance of a solid component on ultrasound, a sharp rise in CA-125 combined with elevated HE4. The full oncological picture is best assessed through a tumour marker panel reviewed by a gynaecological oncologist.
Usually yes. Because the disease is oestrogen-dependent, the fall in oestradiol at menopause suppresses lesion activity. Most women experience significant pain reduction or complete resolution. The exception is women on hormone replacement therapy: exogenous oestrogen can reactivate lesions. Combined oestrogen-progestin HRT (not oestrogen-only) is recommended for these patients. A rare but documented scenario is malignant transformation of a longstanding endometrioma in postmenopause, particularly when new solid components appear on surveillance imaging.
Not currently — there is no definitive cure. Laparoscopic excision reduces pain and improves fertility, but within 5 years of surgery 20–40% of patients experience recurrence. Long-term maintenance hormonal therapy after surgery reduces recurrence rates approximately in half. The goal of treatment is not 'remove and forget' but sustained symptom control, preservation of ovarian reserve and quality of life over the long term.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service