Ovarian Cancer: Symptoms, Diagnosis and CA-125 Tumour Marker
Seven in ten women with ovarian cancer learn their diagnosis at stage III or IV — when the tumour has long since spread beyond the ovary. Not because the disease is undetectable, but because its symptoms are too ordinary: abdominal bloating, pelvic heaviness, early satiety at meals. It is precisely this camouflage behind everyday discomfort that has earned ovarian cancer the name "the silent killer".
What Ovarian Cancer Is and Its Types
Ovarian cancer is a malignant tumour arising from the cells of the ovaries, fallopian tubes or peritoneum. It ranks fourth among cancer deaths in women despite relatively modest incidence — the reason is overwhelmingly late diagnosis.
Epithelial cancer accounts for 90% of all cases and includes several histological subtypes with distinct biological behaviour:
- High-grade serous carcinoma — the most frequent and aggressive subtype, closely linked to BRCA1/2 mutations; often originates from the fimbrial end of the fallopian tube rather than the ovary itself
- Clear cell and endometrioid carcinoma — strongly associated with endometriosis: ectopic endometrial lesions within the ovary undergo malignant transformation in 1–3% of cases
- Mucinous carcinoma — a rare subtype (3–4%); characteristically elevates CEA alongside CA-125, requiring differential diagnosis from metastatic gastrointestinal cancer
Germ cell tumours (5–7%) arise from the egg-producing cells and occur predominantly in young women and adolescents. They are marked by hCG, AFP and LDH. They respond well to chemotherapy — prognosis with timely treatment is favourable even in advanced disease.
Stromal tumours (5%) — granulosa and theca cell tumours; they produce oestrogens, manifesting as uterine bleeding in postmenopausal women or precocious puberty in girls.
FIGO staging determines the extent of surgery and prognosis:
| Stage | Description | 5-year survival |
|---|---|---|
| I | Tumour confined to ovaries or fallopian tubes | 90–92% |
| II | Extension to pelvic organs | 70–80% |
| III | Peritoneal involvement and/or regional lymph nodes | 20–40% |
| IV | Distant metastases (pleura, liver parenchyma) | 5–20% |
Causes and Risk Factors
The exact cause of malignant transformation is unknown, but risk factors are well established. Understanding them matters: some are modifiable, and others allow risk stratification to guide surveillance decisions.
BRCA1 and BRCA2 mutations — the most significant hereditary risk factors. Women with a BRCA1 mutation face a lifetime ovarian cancer risk of 39–46%; with BRCA2 the risk is 10–27%. The population risk, by comparison, is approximately 1–2%. The same mutations dramatically elevate the risk of breast cancer, which is why genetic counselling is essential when family history involves both tumour types.
Lynch syndrome (MMR gene mutations) raises ovarian cancer risk to 8–12%, in addition to its well-known association with colorectal cancer.
Endometriosis triples the risk; the greatest risk is associated with ovarian endometriomas. Chronic inflammation and oxidative stress in ectopic lesions promote the accumulation of oncogenic mutations.
Obesity — a moderate but established factor: BMI above 30 increases risk by 25–30%, particularly for the endometrioid subtype.
Reproductive history. Each pregnancy and period of lactation reduces risk — through fewer lifetime ovulations and the protective action of progesterone. Women who have never been pregnant have 30–60% higher risk. Oral contraceptive use reduces ovarian cancer risk by 30–50% depending on duration of use, with the protective effect persisting for up to 30 years after discontinuation.
Age: 50% of cases are diagnosed in women over 63. Below age 40, ovarian cancer is uncommon and more often a germ cell tumour.
Protective factors: pregnancy, lactation, combined oral contraceptives, tubal ligation and salpingectomy (removal of the fallopian tubes during elective pelvic surgery reduces serous ovarian cancer risk by 40–60%).
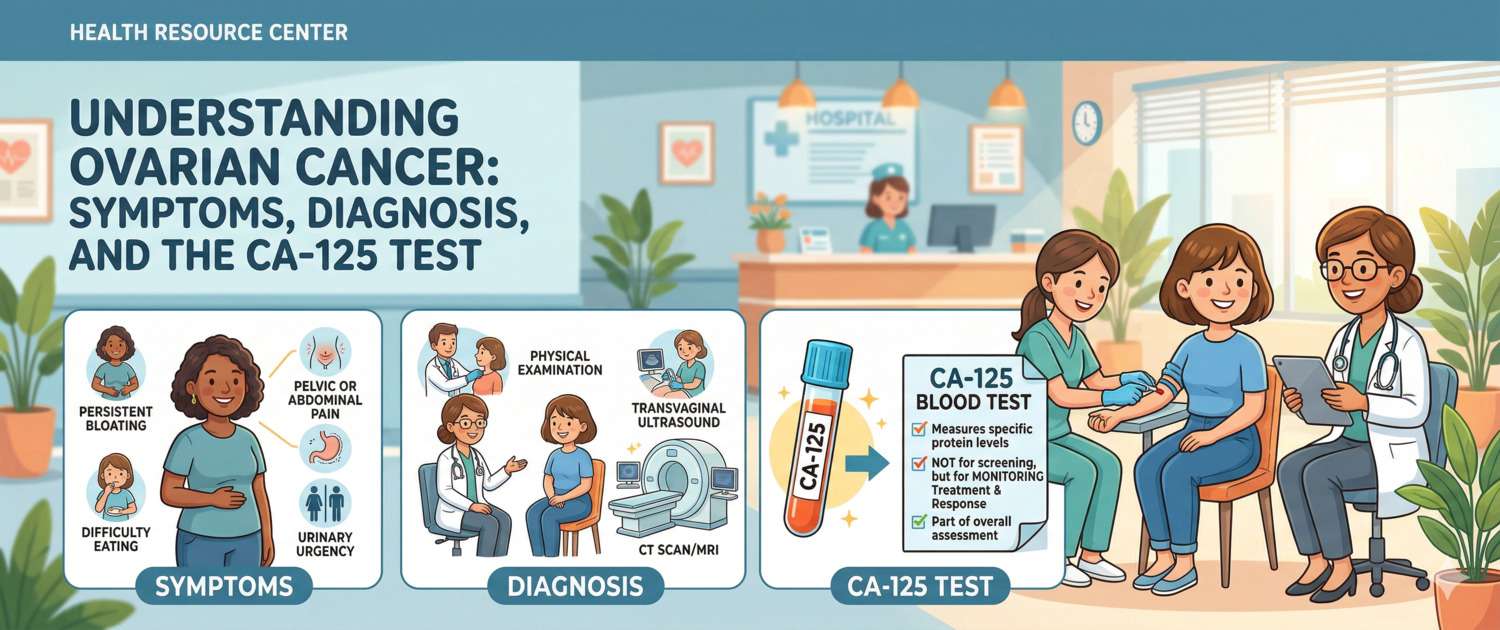
Symptoms: Why Ovarian Cancer Is Silent
The absence of specific early symptoms is the primary driver of high mortality. A tumour growing in the peritoneal cavity causes no urinary, bowel or sexual dysfunction for a prolonged period. The first complaints appear when the tumour reaches a considerable size or ascites — fluid accumulation in the abdomen — develops.
Symptoms that should prompt evaluation:
- Abdominal bloating or visible increase in abdominal girth unrelated to food — particularly when progressive over several weeks
- Persistent sensation of pressure or heaviness in the lower abdomen or pelvis
- Early satiety at meals, reduced appetite
- Increased urinary frequency or a feeling of bladder pressure without an infectious cause
- Unexplained fatigue and weakness
- Change in bowel habit without an obvious cause
- Lower back pain
The treachery of these symptoms lies in their non-specificity: each is readily explained by irritable bowel syndrome, dietary changes or stress. Most women with early ovarian cancer symptoms delay seeking medical attention by five to six months, assuming the cause is trivial.
Urgent warning signs requiring immediate evaluation:
- Sudden severe abdominal pain (possible ovarian torsion or cyst rupture)
- Rapidly progressive abdominal distension over days (rapidly accumulating ascites)
- Several of the above symptoms occurring together and persisting for more than 3 weeks
Diagnosis: Ultrasound, Laboratory Tests and Surgery
Diagnosis relies on the combination of clinical assessment, imaging and laboratory markers. A definitive diagnosis is established only histologically — through biopsy or surgery.
Transvaginal ultrasound is the first-line investigation for any suspected ovarian pathology. Assessment includes size, morphology (cystic, solid, mixed), septal thickness, papillary projections and Doppler vascularity. Features that raise malignancy concern: solid component, thick irregular septa, bilateral disease, ascites.
CT of the abdomen and pelvis with contrast — for staging: assessment of peritoneal disease, lymph nodes and liver metastases. MRI provides additional characterisation in complex cases.
Diagnostic laparotomy is the only method for definitive histological diagnosis and accurate surgical staging. Percutaneous ultrasound-guided biopsy of ovarian masses is contraindicated — it risks peritoneal dissemination of tumour cells, which worsens prognosis.
Laboratory tests:
Complete blood count — detects anaemia in chronic disease. Thrombocytosis (> 400 × 10⁹/L) is a frequent accompaniment of ovarian cancer and an independent adverse prognostic factor: tumour-derived thrombopoietic cytokines drive platelet overproduction.
Tumour marker panel — CA-125 is mandatory when ovarian cancer is suspected; for germ cell tumours, hCG and AFP are added; HE4 is measured alongside CA-125 for ROMA index calculation.
CA-125 Tumour Marker: Capabilities and Limitations
CA-125 (carbohydrate antigen 125) is a glycoprotein produced by serous epithelial cells. The normal upper limit in non-pregnant women is 35 U/mL.
What CA-125 provides:
- Elevated in only 50% of stage I patients — the central argument against its use as a general population screening test
- Elevated in 85–90% of patients at stages III–IV
- After cytoreductive surgery and chemotherapy — the principal instrument for monitoring treatment response and detecting recurrence
- A rising CA-125 after remission precedes clinical recurrence by an average of 3–4 months
Limitations of CA-125: The marker lacks specificity. It rises in endometriosis, pelvic inflammatory disease, pregnancy, liver cirrhosis, pericarditis, pleural effusion and several other malignancies including endometrial, pancreatic and breast cancers. One to two percent of healthy premenopausal women have mildly elevated CA-125 physiologically, and levels rise transiently during menstruation.
ROMA index (Risk of Ovarian Malignancy Algorithm) — a combination of CA-125 + HE4 adjusted for menopausal status. It achieves substantially higher specificity than CA-125 alone, correctly classifying benign from malignant ovarian masses with up to 90% accuracy in postmenopausal women.
IOTA algorithm — an evidence-based ultrasound classification system for ovarian masses; used alongside CA-125 in specialised centres to determine the likelihood of malignancy before surgery.
Treatment of Ovarian Cancer
The cornerstone of treatment is surgery combined with chemotherapy. The extent of the operation and the systemic therapy regimen are determined by stage, histological subtype and molecular characteristics.
Surgery. For early stages (I–II): total hysterectomy with bilateral salpingo-oophorectomy, omentectomy and peritoneal biopsy. For stages III–IV: cytoreductive surgery aiming to remove all visible tumour. Achieving R0 resection (no macroscopic residual disease) is the single most important surgical prognostic factor: in optimally debulked stage III disease, 5-year survival is two to three times higher than in suboptimally debulked patients.
In young women who wish to preserve fertility, a conservative approach removing only the affected ovary is acceptable for stage IA G1 disease.
Chemotherapy. The standard regimen is carboplatin + paclitaxel for 6 cycles. Intraperitoneal chemotherapy in stage III patients with optimal debulking significantly improves survival but is less well tolerated.
PARP inhibitors (olaparib, niraparib) — maintenance therapy after platinum-based chemotherapy. They block DNA repair in tumour cells carrying a homologous recombination defect (BRCA mutations, HRD). Olaparib reduces progression risk by 70% in BRCA-mutant patients; the median progression-free survival at stage III increases from 14 to 56 months with maintenance olaparib.
Bevacizumab (anti-VEGF) is added to chemotherapy at stages III–IV and used as maintenance; it significantly extends time to progression in high-risk subtypes.
At recurrence, treatment strategy is guided by platinum sensitivity: platinum-sensitive relapse (occurring more than 6 months after first-line therapy) responds well to repeated platinum-based regimens; platinum-resistant relapse requires alternative approaches.
When to See a Doctor and Screening
Screening for ovarian cancer is not recommended in the general population: the landmark UKCTOCS trial (200,000 women) demonstrated that neither CA-125 nor transvaginal ultrasound as screening tools reduce mortality. For women at high risk (BRCA1/2 mutations, Lynch syndrome), a surveillance protocol is in place: transvaginal ultrasound plus CA-125 every 6 months from age 25–35. These women are also offered risk-reducing salpingo-oophorectomy after completing childbearing — it reduces ovarian cancer risk by 80% and breast cancer risk by 50% in BRCA1 carriers.
See a gynaecologist promptly when one or more of the following apply:
- progressive abdominal bloating unrelated to diet, persisting more than 3 weeks;
- constant pressure sensation in the lower abdomen or pelvis;
- early satiety and poor appetite occurring alongside other symptoms;
- a palpable mass in the lower abdomen;
- an incidental ovarian mass found on ultrasound performed for any other reason.
If you or close female relatives have had ovarian, breast, colorectal or endometrial cancer — discuss BRCA1/2 and MMR genetic testing with your doctor or a clinical geneticist. Early identification of carrier status transforms the surveillance strategy and makes genuine prevention possible.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
In the general population, screening is not recommended: the UKCTOCS trial (200,000 women) showed that neither CA-125 nor transvaginal ultrasound as screening tools reduce mortality. The main route to early-stage diagnosis today is incidental detection during ultrasound or CT performed for another reason. For women with BRCA1/2 mutations or Lynch syndrome, a specific surveillance protocol exists with considerably better detection rates for early-stage disease.
CA-125 is a non-specific marker: it rises in endometriosis, uterine fibroids, pelvic inflammatory disease, pregnancy, liver cirrhosis and even during menstruation. Approximately one in five women with an elevated CA-125 has no malignant tumour. This is precisely why CA-125 cannot be used as a standalone screening test — it is interpreted only in combination with transvaginal ultrasound findings and the clinical picture.
In 15–20% of cases, yes. BRCA1 and BRCA2 mutations account for the majority of hereditary cases: lifetime risk with BRCA1 reaches 39–46%, with BRCA2 it is 10–27%. Lynch syndrome (MMR gene mutations) also elevates ovarian cancer risk and is simultaneously the principal cause of hereditary colorectal cancer. When two or more first-degree female relatives have had ovarian or breast cancer, genetic counselling and BRCA testing are warranted.
Prognosis depends fundamentally on stage and subtype. When detected at stage I, five-year survival exceeds 90%. At stage III — where most patients are diagnosed — it is 20–40%. Modern PARP inhibitors have substantially improved outcomes in BRCA-mutant patients: median progression-free survival at stage III with olaparib maintenance increased from approximately 14 to 56 months in the landmark SOLO-1 trial, compared with placebo.
Yes — and protectively. Combined oral contraceptive use reduces ovarian cancer risk by 30–50%: the longer the duration of use, the stronger the effect. Protection persists for up to 30 years after discontinuation. The mechanism involves suppression of ovulation and reduction in gonadotrophin-driven stimulation of ovarian epithelium. This is especially relevant for BRCA mutation carriers: oral contraceptives reduce their elevated baseline risk, though they do not eliminate it.
Carboplatin and paclitaxel carry moderate hepatotoxicity. Liver function tests — ALT, AST, bilirubin and alkaline phosphatase — are checked before each treatment cycle. Mild transaminase elevation (up to 2–3× the upper limit of normal) is common in early cycles and does not require treatment discontinuation. More pronounced or rising values prompt dose modification. In patients with hepatic metastases, monitoring is particularly important: tumour response to treatment can itself cause a transient rise in liver enzymes as lesions regress.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service