Breast Cancer: Symptoms, Diagnosis and Treatment Guide
One in eight women globally will develop breast cancer during her lifetime. It is the most common cancer in women — and simultaneously one of the most treatable when found early: five-year survival at stage I exceeds 95%. The gap between that figure and real-world mortality comes down to one thing — late diagnosis. Understanding symptoms, risk factors and screening principles is what genuinely saves lives.
What Breast Cancer Is and Its Molecular Subtypes
Breast cancer is a malignant tumour arising from the glandular tissue or ducts of the breast. In 80–85% of cases it is invasive ductal carcinoma (IDC) — a tumour from duct cells invading surrounding tissue. Lobular carcinoma (ILC, 10–15%) arises from the lobules and is often found simultaneously in both breasts.
Modern oncology classifies breast cancer primarily by molecular subtype rather than histological type — this determines treatment selection:
| Subtype | Receptor profile | Proportion | Prognosis |
|---|---|---|---|
| Luminal A | ER+/PR+, HER2–, low Ki-67 | 40–45% | Best |
| Luminal B | ER+/PR±, HER2– or HER2+, high Ki-67 | 20–25% | Intermediate |
| HER2-enriched | ER–/PR–, HER2+ | 10–15% | Improved with targeted therapy |
| Triple negative | ER–/PR–, HER2– | 15–20% | Most aggressive |
Luminal subtypes are hormone-dependent: oestradiol and progesterone stimulate their growth, which is why anti-oestrogenic therapy is central to their treatment. HER2-enriched tumours depend on HER2 receptor overactivation — specific targeted agents have transformed their prognosis. Triple-negative breast cancer (TNBC) lacks all three receptors and is the most challenging to treat: aggressive, more frequent in young women and BRCA1 mutation carriers.
TNM staging:
| Stage | Description | 5-year survival |
|---|---|---|
| 0 | Carcinoma in situ (DCIS, LCIS) | ~99% |
| I | Tumour ≤ 2 cm, no nodal involvement | 95–99% |
| II | Tumour 2–5 cm or 1–3 positive axillary nodes | 75–90% |
| III | Locally advanced disease | 40–70% |
| IV | Distant metastases (bone, lung, liver, brain) | 20–30% |
Causes and Risk Factors
Breast cancer has well-characterised risk factors. Approximately 30% of all cases are potentially linked to modifiable lifestyle factors.
BRCA1 and BRCA2 mutations — the most significant hereditary risk factors. Lifetime breast cancer risk with BRCA1 reaches 70–80%; with BRCA2, 45–85%. The same mutations dramatically raise the risk of ovarian cancer, which is why carriers are offered either prophylactic bilateral mastectomy or an intensive surveillance protocol. Other high-risk genes include PALB2 (35–60% risk), CHEK2, ATM and TP53 (Li–Fraumeni syndrome — risk up to 85%).
Age and reproductive history. Risk rises steeply after age 40 and accelerates after 50. Early menarche (before 12), late menopause (after 55) and nulliparity increase cumulative oestrogen exposure to breast tissue, raising risk by 20–50%.
Hormone replacement therapy (HRT): combined oestrogen-progestogen HRT used for more than 5 years raises breast cancer risk by 20–25%. Oestrogen-only therapy in women after hysterectomy carries considerably lower risk and only when used for more than 10 years.
Obesity after menopause: adipose tissue is the primary source of oestrogen in postmenopause through peripheral aromatisation of androgens. BMI above 30 raises postmenopausal breast cancer risk by 30–60%. Before menopause, obesity modestly reduces luminal subtype risk while raising triple-negative risk.
Alcohol: one of the few well-established dietary risk factors. Each 10 g of alcohol per day raises breast cancer risk by 7–10%, mediated through elevated circulating oestradiol.
Breast tissue density: high mammographic density (BI-RADS category D) increases risk fourfold to sixfold and simultaneously impairs tumour detection on mammography — supplemental ultrasound or MRI is needed.
Prior chest radiotherapy in young age (for example, for Hodgkin's lymphoma) — a significant risk factor: breast cancer may develop 15–30 years after irradiation.
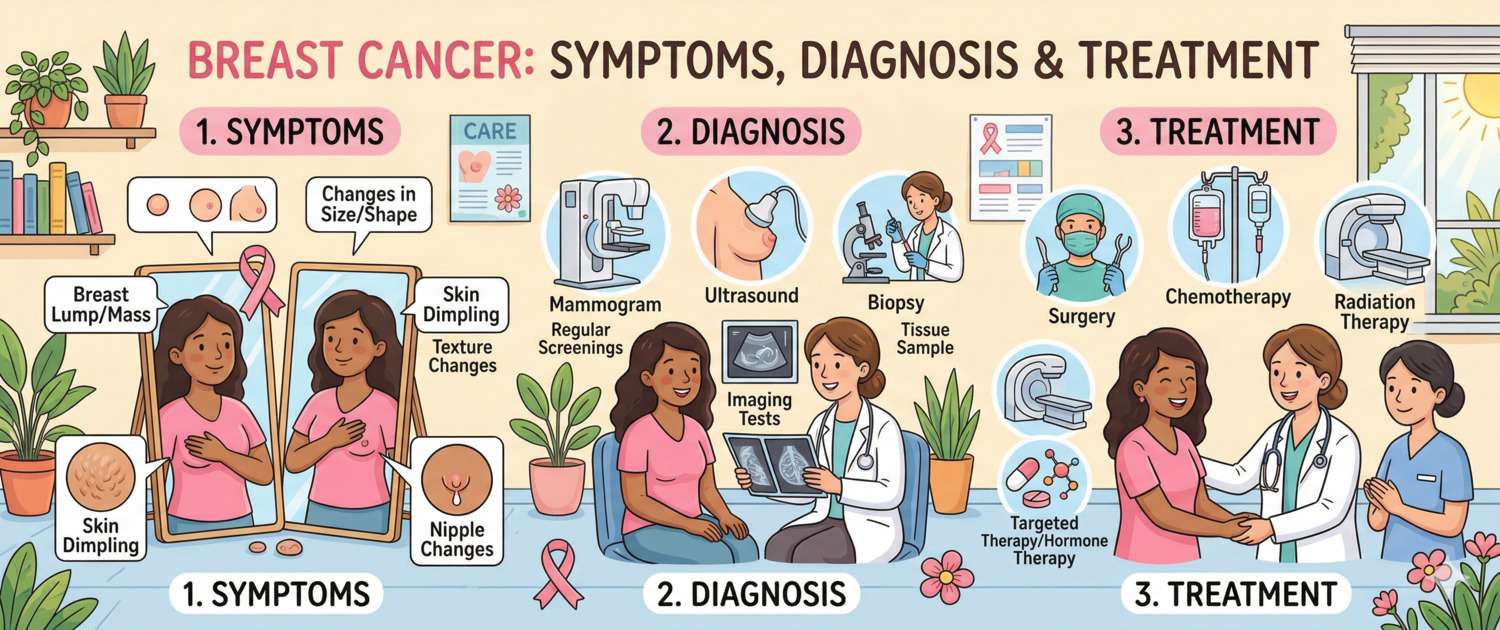
Symptoms: Early and Late Signs
Early breast cancer often has no symptoms and is detected only on mammography — which is precisely why regular screening is so important. When symptoms appear, this often indicates a more advanced process.
Signs requiring prompt medical evaluation:
- A new lump or thickening in the breast or axilla that was not previously present. Most palpable lumps are benign (cysts, fibroadenomas), but any new finding requires assessment
- A change in breast shape or size
- Peau d'orange — diffuse skin thickening and oedema resembling the surface of an orange. A sign of lymphovascular invasion — characteristic of inflammatory breast cancer
- Skin or nipple retraction — a sign of tethering by Cooper's ligaments
- Nipple discharge — particularly bloody or from one breast only
- Redness, weeping or ulceration of the areola or nipple — a sign of Paget's disease of the breast
- Persistent swelling, redness and warmth of the entire breast without signs of infection — inflammatory breast cancer requiring urgent evaluation
Breast pain is not a characteristic symptom of breast cancer. Most painful lumps prove to be benign. However, the absence of pain is not a marker of benignity — most malignant tumours are painless at early stages.
Diagnosis: Mammography, Biopsy and Blood Tests
Diagnosis proceeds through three levels: screening investigation → confirmatory imaging → morphological verification by biopsy.
Mammography — the gold standard for screening in women from age 40–50. Digital two-projection mammography detects tumours of 5–10 mm, inaccessible to palpation. Digital breast tomosynthesis (3D mammography) reduces false-positive rates in women with dense breast tissue.
Breast ultrasound — supplementary for dense tissue, women under 40, palpable masses and to differentiate cystic from solid lesions.
Contrast-enhanced breast MRI — the most sensitive method (95%). Indicated for: BRCA1/2 carriers; indeterminate mammography/ultrasound findings; extent assessment before breast-conserving surgery; lobular carcinoma (frequently multifocal).
Biopsy — the only method of morphological diagnosis. Core-needle biopsy under ultrasound or stereotactic guidance is the standard: it provides tissue for histology and immunohistochemistry (ER, PR, HER2, Ki-67). Vacuum-assisted biopsy is used for microcalcifications.
Laboratory tests once diagnosis is confirmed:
Complete blood count — baseline haematopoietic status before chemotherapy; neutropenia and anaemia are the most common dose-limiting toxicities of cytotoxic therapy.
Liver function tests — when hepatic metastases are suspected and for monitoring hepatotoxicity of targeted agents (trastuzumab, lapatinib).
Tumour marker panel — CA 15-3 and CEA are used for monitoring in metastatic breast cancer: rising levels during remission are an early sign of progression. Not used for primary diagnosis.
Molecular Diagnostics: Receptor Status and Mutations
The pathology report from biopsy is the most important document in breast cancer management. Beyond histological type, it contains:
Receptor status: ER (oestrogen receptor) and PR (progesterone receptor) determine sensitivity to hormonal therapy. ER positivity (≥ 1% staining) indicates eligibility for anti-oestrogenic treatment. High expression (Allred score ≥ 6) predicts maximum benefit.
HER2 status: determined by immunohistochemistry (3+ = positive; 2+ = equivocal → FISH/CISH for clarification). HER2 positivity is the indication for trastuzumab, pertuzumab and other targeted agents. The emerging category "HER2-ultralow" (1+) is a potential target for trastuzumab deruxtecan.
Ki-67: proliferation index. Ki-67 above 20–30% — high proliferative activity, an adverse prognostic factor and indication for more intensive systemic therapy.
Genomic tests (Oncotype DX, MammaPrint) — in early-stage luminal disease, calculate the risk of distant recurrence and guide the decision on chemotherapy. In low-genomic-risk patients, chemotherapy does not improve survival — allowing avoidance of its toxicity.
BRCA1/2 testing is indicated in all patients under 50, triple-negative breast cancer at any age, and when family history includes breast or ovarian cancer. Identifying a mutation changes systemic treatment strategy (PARP inhibitors) and determines surveillance for the contralateral breast.
Treatment of Breast Cancer
Breast cancer treatment is multidisciplinary: surgeon, oncologist, radiologist, reconstructive surgeon and psychologist work as a team. Strategy is determined by stage, molecular subtype, receptor and HER2 status.
Surgery:
- Breast-conserving surgery (lumpectomy/wide local excision) followed by radiotherapy — for tumours ≤ 3–4 cm without multifocality. Equivalent to mastectomy in overall survival when indications are met
- Mastectomy — for multifocal disease, contraindications to radiotherapy, BRCA carriership or patient preference. Immediate reconstruction is the standard in modern centres
- Sentinel lymph node biopsy has replaced routine axillary lymph node dissection when nodes are clinically uninvolved, dramatically reducing lymphoedema rates with equivalent oncological outcomes
Radiotherapy: after breast-conserving surgery — mandatory. After mastectomy — when ≥ 4 nodes are involved, tumour is large or resection margins are positive.
Chemotherapy:
- Neoadjuvant (before surgery): shrinks the tumour for breast conservation and assesses in vivo response. Achieving pathological complete response (pCR) significantly improves prognosis
- Adjuvant (after surgery): standard for HER2+ and TNBC; in luminal subtypes — guided by genomic testing and stage
- Regimens: anthracyclines (doxorubicin, epirubicin) ± taxanes (paclitaxel, docetaxel) — the foundation of most protocols
Hormonal (endocrine) therapy — only for ER+/PR+ tumours:
- Tamoxifen (SERM): blocks oestrogen receptors in breast tissue; standard for premenopausal women; 5–10 years reduces recurrence risk by 40–50%
- Aromatase inhibitors (letrozole, anastrozole, exemestane): block peripheral oestradiol synthesis; outperform tamoxifen in postmenopausal women
- CDK4/6 inhibitors (palbociclib, ribociclib, abemaciclib) + aromatase inhibitor — first-line standard for metastatic ER+/HER2– breast cancer: approximately double median progression-free survival
HER2-targeted therapy:
- Trastuzumab (Herceptin) — in adjuvant setting reduces recurrence risk by ~50% in HER2+ tumours; 12 months
- Pertuzumab — added to trastuzumab at high risk; synergistic effect
- Trastuzumab deruxtecan (T-DXd) — antibody-drug conjugate; has transformed metastatic HER2+ and HER2-low breast cancer outcomes: median progression-free survival in DESTINY-Breast04 was 9.9 versus 5.1 months with chemotherapy
PARP inhibitors (olaparib, talazoparib): in metastatic breast cancer with germline BRCA1/2 mutations — significantly improve progression-free survival compared with chemotherapy.
Immunotherapy: pembrolizumab + chemotherapy — first-line standard for metastatic PD-L1-positive TNBC; in the neoadjuvant setting for high-risk early TNBC, increases pCR rates.
Screening and Prevention
Mammographic screening:
- Average risk: annually or every 2 years from age 40 (NCCN, ACR) or 50 (some European guidelines) to age 74–75; the optimal starting age is debated across countries — confirm with your gynaecologist
- High risk (BRCA, PALB2, lifetime risk > 20%): annual MRI + mammography from age 25–30
Breast self-examination: monthly on days 5–7 of the cycle. Does not replace mammography, but allows changes to be noticed between screening visits. Familiarity with the normal look and feel of one's breasts is a valuable skill.
Prevention:
- Maintain healthy weight — particularly after menopause
- Limit alcohol to ≤ 1 drink per day or abstain
- Regular physical activity (≥ 150 minutes moderate-intensity per week reduces risk by 10–20%)
- Breastfeeding ≥ 12 cumulative months — reduces risk by 10–15%
- For BRCA1/2 carriers — discuss prophylactic bilateral mastectomy: reduces breast cancer risk by ~95%
When to See a Doctor
Consult a breast surgeon or gynaecologist promptly when any of the following apply:
- a new lump or thickening in the breast or axilla;
- change in breast shape, size or symmetry;
- skin or nipple retraction, or peau d'orange;
- bloody or unilateral nipple discharge;
- persistent redness, swelling or warmth of the breast without infection.
If you are over 40 and have not had a mammogram in more than two years — arrange screening without waiting for symptoms. With a family history of breast or ovarian cancer, discuss BRCA1/2 testing and an individualised surveillance protocol with your doctor.
Early breast cancer is highly treatable. Five-year survival at stage I is 95–99%. Do not delay evaluation — the cost of postponement in this diagnosis is measured in stage, and therefore in treatment options.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
Prognosis is determined by stage, molecular subtype and access to modern treatment. At stage I, five-year survival exceeds 95%; at stage II, 75–90%; at stage III, 40–70%; at stage IV, approximately 25–30% with modern targeted therapy — and for some subtypes (ER+/HER2–), median overall survival in metastatic disease now exceeds 5 years. Triple-negative cancer carries the worst stage-for-stage prognosis in early disease, but patients who achieve complete pathological response to chemotherapy have outcomes comparable to luminal subtypes.
Recommendations vary by country. Most modern guidelines (NCCN, ACR) recommend annual mammography from age 40 for average-risk women. Several European standards recommend biennial screening from age 50. Women at elevated risk (family history, dense breast tissue, prior atypical hyperplasia on biopsy) should begin earlier. BRCA1/2 carriers: annual MRI plus mammography from age 25–30. Confirm the optimal starting age for your individual risk profile with a gynaecologist or breast specialist.
Combined HRT (oestrogen plus progestogen) used for more than 5 years raises breast cancer risk by 20–25%. Oestrogen-only HRT in women after hysterectomy carries a substantially lower risk and only after more than 10 years of use. The decision about HRT requires a careful individual risk-benefit balance — the same hormones that slightly raise breast cancer risk reduce cardiovascular and bone risks. HRT also increases the risk of deep vein thrombosis, another factor that clinicians weigh when prescribing.
Chemotherapy for breast cancer predictably suppresses bone marrow function. The most common changes are neutropenia (the main infection risk), falling haemoglobin (causing fatigue and breathlessness) and thrombocytopaenia. The nadir — lowest blood count values — occurs on days 7–14 after each cycle. This is why a full blood count is checked before every cycle: neutrophils below 1.5 × 10⁹/L prompt treatment delay. G-CSF (granulocyte colony-stimulating factors) reduces the risk of severe neutropenia and helps maintain treatment intensity on schedule.
Approximately 5–10% of all breast cancer cases are linked to inherited mutations — principally BRCA1 and BRCA2. A further 15–20% show family clustering without an identified syndrome. Indications for genetic testing include: breast cancer before age 50; triple-negative breast cancer at any age; two or more first-degree relatives with breast cancer; ovarian cancer in the family history; male breast cancer in a relative. Identifying a mutation shapes not only the patient's own treatment but also surveillance decisions for her daughters and sisters.
Yes, when appropriate criteria are met. Breast-conserving surgery (lumpectomy with radiotherapy) provides equivalent overall survival to mastectomy in randomised trials when indications are followed. Requirements: tumour ≤ 3–4 cm, single focus, no contraindication to radiotherapy, patient preference. For larger tumours, neoadjuvant chemotherapy can shrink the tumour sufficiently to make conservation feasible. In BRCA1/2 carriers, the risk of a new cancer in remaining breast tissue is higher, which often leads to a decision for bilateral mastectomy.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service