Colorectal Cancer: Symptoms, Diagnosis and Tumour Markers
Most people in whom colorectal cancer is found at an early stage had not suspected it for a single day. The tumour grew silently — without pain, without blood in the stool, without any alarming signal — until a colonoscopy or a stool test detected it during routine screening. This is precisely why colorectal cancer ranks second globally in cancer mortality: not because it is incurable, but because it is found far too late, far too often.
What Colorectal Cancer Is and Its Stages
Colorectal cancer (CRC) is a malignant tumour arising from the epithelial lining of the large bowel or rectum. In 95% of cases it is an adenocarcinoma — a tumour originating from glandular cells. Approximately 60% arise in the colon and 40% in the rectum. This distinction matters for treatment, as rectal cancer requires a specific approach due to the anatomical constraints of the pelvis.
The majority of colorectal cancers develop from adenomatous polyps — benign growths of the mucosal lining. The progression from normal epithelium through a polyp to cancer takes on average 10–15 years, and it is this long window that makes screening so effective: a detected and removed polyp is a prevented cancer.
TNM staging determines treatment strategy and prognosis:
| Stage | Description | 5-year survival |
|---|---|---|
| I | Tumour confined to bowel wall, no nodes | ~90% |
| II | Through the wall, no nodal involvement | 75–85% |
| III | Regional lymph node involvement | 40–70% |
| IV | Distant metastases (liver, lungs) | 10–15% |
More than 1.9 million new cases of CRC are diagnosed worldwide each year. Incidence is rising among people under 50 — increasing by roughly 50% in young adults over the past two decades, making the revision of recommended screening age increasingly urgent.
Causes and Risk Factors
Colorectal cancer is a disease in which lifestyle plays no smaller a role than genetics. Up to 55% of all cases are potentially preventable through modification of known risk factors.
Age is the dominant independent risk factor: 90% of cases are diagnosed in people over 50. Yet over the past 20 years, rates in 20–50-year-olds have risen steadily — attributed in large part to the global obesity epidemic and changes in dietary habits in younger generations.
Diet. Red meat (beef, pork, lamb) and especially processed meat (sausages, hot dogs, bacon, ham) are documented risk factors. Processed meat is classified by the WHO as a Group 1 carcinogen for colorectal cancer. Each additional 50 g of processed meat per day increases risk by 18%. Conversely, a diet high in fibre from wholegrains, legumes, vegetables and fruit reduces risk by 25–40%.
Obesity raises colorectal cancer risk by 30–35%, especially colon cancer in men. Visceral adipose tissue drives chronic inflammation and hyperinsulinaemia — both promote intestinal epithelial proliferation.
Type 2 diabetes and insulin resistance are independent risk factors: chronically elevated insulin and IGF-1 stimulate tumour cell growth through insulin receptors.
Inflammatory bowel disease: ulcerative colitis and Crohn's disease markedly increase risk — after more than 10 years of extensive colitis, risk is 5–10 times higher than in the general population. Chronic mucosal inflammation creates conditions for the accumulation of oncogenic mutations.
Alcohol and smoking: moderate to heavy alcohol intake raises risk by 40–50%. Smoking is most strongly linked to cancers of the right colon.
Hereditary syndromes: Lynch syndrome (hereditary non-polyposis CRC) accounts for 3–5% of all cases; lifetime risk in carriers reaches 70–80%. Familial adenomatous polyposis (FAP) — more than 100 polyps with near 100% malignant transformation without prophylactic colectomy. A positive family history without an identifiable syndrome doubles risk.
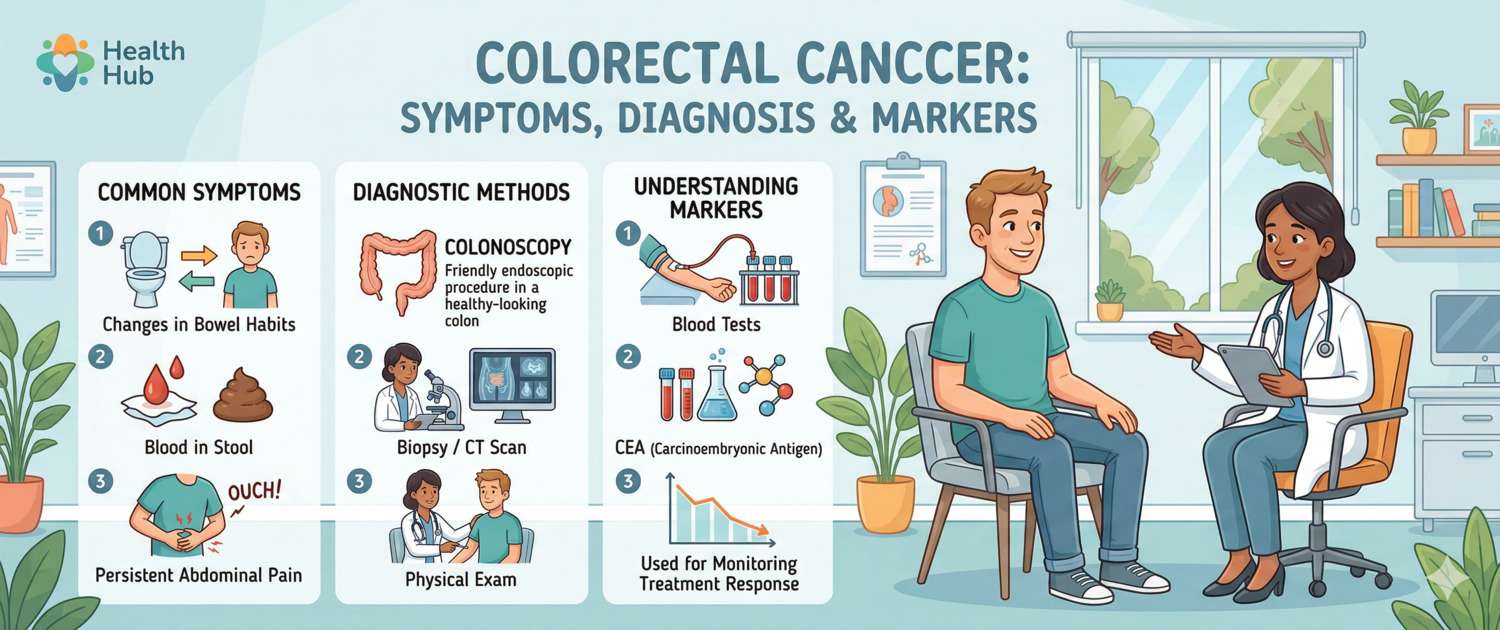
Symptoms: Early and Late Signs
The insidious nature of colorectal cancer lies in its silence: early-stage tumours produce essentially no symptoms as they grow inside the bowel lumen for years before disrupting bowel function or causing visible bleeding.
Symptoms that require prompt medical evaluation:
- Blood in the stool or on toilet paper — even once. Do not attribute it to haemorrhoids without investigation: haemorrhoids and rectal cancer can coexist
- Persistent change in bowel habit lasting 3 weeks or more: alternating constipation and diarrhoea, or a change in the calibre of stools (pencil-thin stools)
- Unexplained weight loss — 5% or more of body weight over 6 months
- Persistent abdominal discomfort or pain, a sensation of incomplete bowel emptying (tenesmus)
- Progressive fatigue and weakness — often the only symptom of occult intestinal bleeding and worsening anaemia
Anaemia is one of the most common presentations of right-sided colon cancer. Tumours in the right colon bleed slowly and invisibly: blood mixes with stool and cannot be seen, but gradually depletes haemoglobin stores. The patient arrives at a haematologist with iron deficiency anaemia in an adult without a clear explanation — this is an absolute indication for colonoscopy.
Diagnosis: Colonoscopy and Blood Tests
Colonoscopy is the gold standard for diagnosis and screening. It is the only method that visualises the entire large bowel, identifies polyps and early tumours, and removes found polyps in the same session. Sensitivity for cancer reaches 95%; for polyps > 1 cm, 85–90%.
Alternative screening methods when colonoscopy is refused or contraindicated:
- Faecal immunochemical test (FIT) — annually; high specificity for human haemoglobin, no dietary restrictions required
- Stool DNA test (Cologuard) — every 3 years; detects tumour-derived DNA and haemoglobin
- CT colonography (virtual colonoscopy) — every 5 years; polyps > 6 mm found on CT require conventional colonoscopy for biopsy
Laboratory tests complement imaging:
Complete blood count — detects hypochromic microcytic anaemia as an indirect sign of chronic intestinal bleeding. Normocytic anaemia may occur in metastatic disease.
Liver function tests — when hepatic metastases are suspected: elevated bilirubin, alkaline phosphatase and ALT point to parenchymal involvement. LDH rises with extensive metastatic disease as a marker of tumour necrosis.
Tumour marker panel — includes CEA and CA 19-9 as the principal markers for post-treatment monitoring and response assessment.
Tumour Markers in Colorectal Cancer
Tumour markers in CRC have a specific role: they are not intended for primary diagnosis, but are indispensable for monitoring after treatment.
CEA (carcinoembryonic antigen) — the principal CRC marker. Normal in non-smokers < 3 ng/mL, in smokers < 5 ng/mL. The clinical logic of CEA:
- Before surgery: baseline level. High CEA (> 10 ng/mL) correlates with later stage and worse prognosis
- After curative resection: should normalise within 4–6 weeks. Persistent elevation indicates incomplete resection or occult metastases
- Surveillance: a rising CEA after normalisation is an early sign of recurrence, often preceding CT detection by 3–6 months
- CEA is not used for primary diagnosis: its sensitivity is only 36–50%, and in 30% of patients with early CRC it remains normal
CA 19-9 as an additional marker is useful in patients with normal CEA despite confirmed tumour, and for differential diagnosis versus pancreatic cancer when hepatic metastases are present. Full details are in the CA 19-9 tumour marker article.
False-positive CEA elevation occurs in hepatic insufficiency, cirrhosis, inflammatory bowel disease and smoking — interpretation always requires clinical context.
Treatment of Colorectal Cancer
Treatment selection is driven by stage, tumour location and molecular characteristics (RAS and BRAF mutation status, MSI status).
Surgery is the foundation of treatment for stages I–III. Right-sided cancers are treated by right hemicolectomy; left-sided by left hemicolectomy or sigmoid resection. Rectal cancers are treated by anterior resection with sphincter preservation or, for low rectal tumours, abdominoperineal resection. Laparoscopic procedures for CRC achieve equivalent oncological outcomes to open surgery with fewer complications.
Chemotherapy for stage III (adjuvant) and stage IV (palliative or conversion):
- FOLFOX (oxaliplatin + 5-fluorouracil + leucovorin) — standard adjuvant therapy for stage III; reduces recurrence risk by 20–25%
- FOLFIRI (irinotecan + 5-FU + leucovorin) — for metastatic disease, particularly when oxaliplatin is not tolerated
Targeted therapy in metastatic CRC:
- Bevacizumab (anti-VEGF) — added to chemotherapy regardless of molecular subtype; improves overall survival
- Cetuximab / panitumumab (anti-EGFR) — only for RAS (KRAS, NRAS) wild-type and BRAF wild-type tumours; RAS mutation is an absolute contraindication to these agents
Immunotherapy: pembrolizumab and other PD-1 inhibitors are effective in MSI-H/dMMR tumours — approximately 15% of all CRC. In these patients, immunotherapy delivers substantially better outcomes than standard chemotherapy.
Radiotherapy is used primarily in rectal cancer: preoperative chemoradiation reduces tumour size and lowers the risk of local recurrence.
Screening and Prevention: How to Detect Cancer Early
Colorectal cancer screening is one of the few medical interventions with a proven mortality reduction: regular screening programmes lower CRC mortality by 30–40%. The mechanism is straightforward — detecting and removing precancerous polyps interrupts the polyp-to-cancer chain.
Who should screen and when:
- Average risk (no family history, no IBD, no hereditary syndrome): colonoscopy from age 45, repeated every 10 years if no pathology found. Alternative: annual FIT test
- Increased risk (one first-degree relative with CRC or advanced adenoma below age 60, or two first-degree relatives): start at 40 or 10 years before the affected relative's age, colonoscopy every 5 years
- High risk (Lynch syndrome, FAP, IBD): individual protocol, often from age 20–25
Prevention: reduce red and processed meat consumption; increase dietary fibre from wholegrains, legumes and vegetables; maintain a healthy body weight; physical activity ≥ 150 minutes of moderate intensity per week reduces CRC risk by 20–30%; limit alcohol; avoid smoking. Aspirin reduces CRC risk in Lynch syndrome carriers, but is not recommended routinely in the general population due to gastrointestinal bleeding risk.
When to See a Doctor
Consult a GP or gastroenterologist promptly when any of the following applies:
- blood in the stool or dark, tarry stools;
- change in bowel habit lasting more than 3 weeks;
- unexplained weight loss exceeding 5% over 6 months;
- iron deficiency anaemia in a man or a postmenopausal woman without a clear cause;
- progressive weakness and pallor without an established diagnosis;
- you are aged 45 or over and have never had CRC screening.
Early colorectal cancer is highly treatable. Five-year survival when detected at stage I exceeds 90%. At stage IV it falls to around 10–15%. The difference between these figures is the value of screening. Do not wait for symptoms before arranging a colonoscopy — by the time symptoms appear, the window for early treatment may already have closed.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
Progressive fatigue and breathlessness on exertion — these can be the only manifestation of chronic intestinal bleeding causing anaemia, particularly with right-sided tumours. Right colon cancers bleed slowly; the blood is invisible in the stool, but haemoglobin falls steadily over months. Many patients are treated for 'chronic anaemia' for years before colonoscopy reveals the cause. The second underappreciated symptom is a subtle change in stool calibre with sigmoid cancer — patients attribute it to diet and seek no investigation.
CEA is not used for primary diagnosis — its accuracy for this purpose is insufficient. In 30–40% of patients with early colorectal cancer, CEA remains within the normal range. Conversely, it can be mildly elevated in 15–20% of healthy individuals — particularly smokers, those with liver disease or inflammatory bowel disease. CEA's primary clinical value is post-treatment surveillance: a rising level after normalisation warns of recurrence 3–6 months before it becomes visible on CT scanning. Diagnosis requires colonoscopy with biopsy.
For people at average risk, most guidelines now recommend beginning screening at age 45: colonoscopy every 10 years if no pathology is found, or an annual faecal immunochemical test (FIT). With a family history of CRC in a first-degree relative below age 60, start at 40 or 10 years before the affected relative's age of diagnosis. Lynch syndrome and FAP require an individual protocol under specialist supervision, typically beginning in the mid-twenties.
In 5–10% of cases, yes — through recognised hereditary syndromes: Lynch syndrome (MMR gene mutations) and familial adenomatous polyposis (APC mutation). A further 20–25% of cases show a familial clustering without an identified syndrome, likely mediated through polygenic variants. When two or more first-degree relatives have had CRC, or one has had it before age 60, genetic counselling and earlier screening are warranted.
Stage IV is considered incurable in most cases, but treatment significantly extends life and improves its quality. A subset of patients with isolated liver or lung metastases is eligible for surgical resection with potentially curative intent — 5-year survival after complete resection of liver metastases reaches 20–40%. Modern chemotherapy combined with targeted agents and immunotherapy in MSI-H tumours achieves a median overall survival of 30–36 months at stage IV, compared with 6–12 months in the pre-targeted therapy era.
Substantially. Processed meat (sausages, bacon, ham) is classified by the WHO as a Group 1 carcinogen for CRC: each 50 g daily increment raises risk by 18%. Red meat is a probable carcinogen (Group 2A). Dietary fibre from wholegrains, legumes and vegetables reduces risk by 25–40%, likely through accelerated bowel transit and beneficial changes in the gut microbiome. Regular alcohol intake raises risk. Checking liver function tests alongside a full metabolic assessment is a reasonable starting point when multiple dietary risk factors are present.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service