Type 1 Diabetes: Symptoms, Diagnosis and Treatment
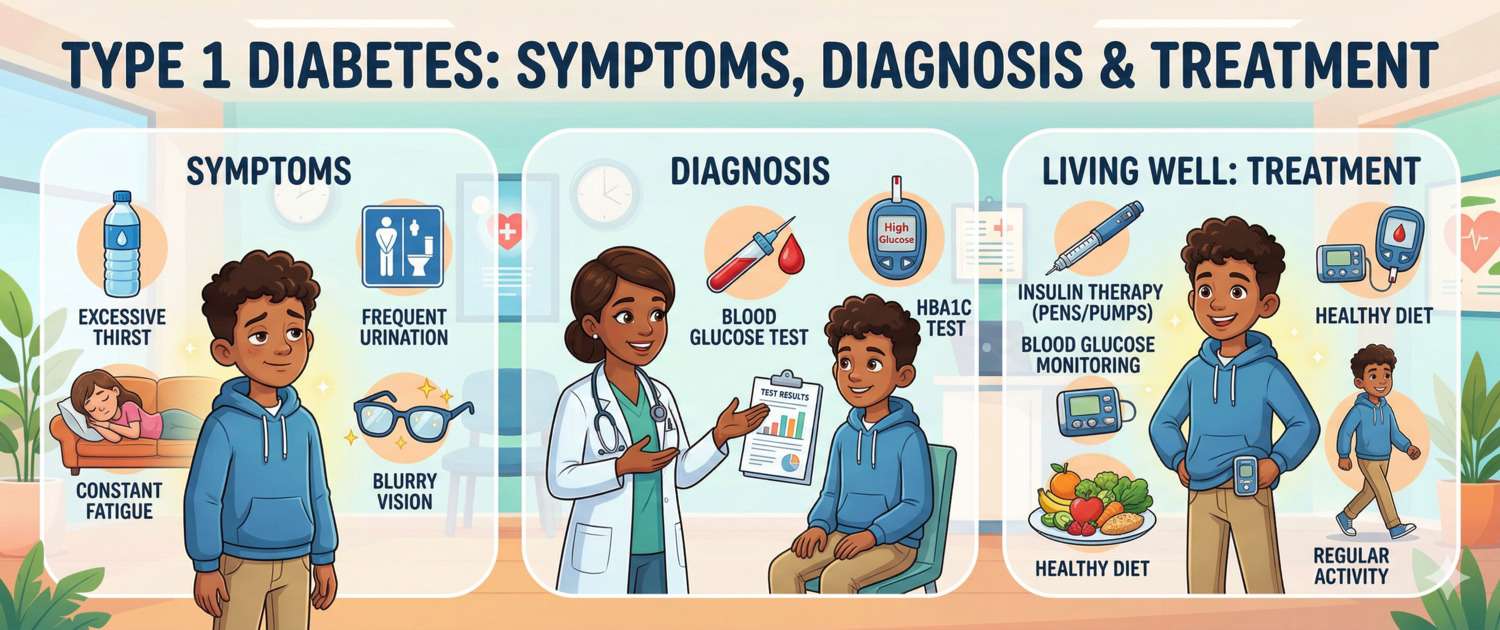
Type 1 diabetes is a disease that changes life in a single day. Yesterday a young person was healthy; today — thirst, frequent urination, rapid weight loss and, if the moment is missed, ketoacidosis and intensive care. Unlike type 2 diabetes, no diet will help and there is no "wait and see" period: the β-cells are gone, and insulin is needed from day one — for life.
What Type 1 Diabetes Is
Type 1 diabetes mellitus (T1DM) is a chronic autoimmune disease in which the immune system destroys the insulin-producing β-cells of the islets of Langerhans in the pancreas. When 80–90% of β-cells are lost, absolute insulin deficiency develops and clinical diabetes manifests.
Insulin is the key anabolic hormone: it unlocks cellular access to glucose. Without it, glucose accumulates in the blood (hyperglycaemia) while cells "starve." In response, the body switches to fat breakdown → ketone bodies are produced → metabolic acidosis develops (ketoacidosis) — a direct threat to life.
Epidemiology: T1DM accounts for 5–10% of all diabetes cases. Incidence is highest in children and adolescents (peak age 10–14 years), but T1DM can manifest at any age, including adults (LADA — latent autoimmune diabetes of adults). Incidence varies considerably by country and ethnicity.
Genetics and triggers: 90% of T1DM patients carry HLA-DR3 or HLA-DR4 haplotypes. Concordance in identical twins is only 30–50%, confirming that external triggers are required: viral infections (Coxsackievirus B), microbiome changes, early exposure to cow's milk protein. The precise mechanism initiating the autoimmune attack remains incompletely understood.
Onset Symptoms: When Time Cannot Be Wasted
The classic "four Ps" of T1DM presentation:
Polyuria — frequent, high-volume urination including at night. Mechanism: hyperglycaemia → glucose filtered into urine → osmotic diuresis → fluid and electrolyte loss.
Polydipsia — unquenchable thirst in response to fluid loss.
Weight loss — rapid loss (5–10 kg or more over a few weeks) without reduced appetite. Mechanism: cells cannot access glucose → catabolism of protein and fat.
Polyphagia — increased appetite despite ongoing weight loss (the paradox of "hunger amid plenty").
Additional symptoms: fatigue and weakness, blurred vision (from osmotic lens changes in hyperglycaemia), slow wound healing, fungal infections (candidiasis).
In children, onset is often rapid — progressing to full ketoacidosis within days. Secondary nocturnal enuresis in a previously toilet-trained child is a warning sign.
LADA (in adults): onset after age 30, frequently misclassified as T2DM. Distinguishing features: lean patient, rapid insulin requirement, positive autoantibodies (GADA, IA-2A).
Diagnosis of Type 1 Diabetes
Diagnostic criteria for diabetes (WHO)
Diabetes is diagnosed when at least one of the following is present:
- Fasting glucose ≥ 7.0 mmol/L (on two occasions)
- 2-hour glucose ≥ 11.1 mmol/L on OGTT
- Random glucose ≥ 11.1 mmol/L + symptoms
- HbA1c ≥ 6.5% (on two occasions)
With classic symptoms and random glucose ≥ 11.1 mmol/L, repeat confirmation is not required.
Specific markers of T1DM
Autoantibodies — confirm autoimmune aetiology:
- GADA (glutamic acid decarboxylase antibodies) — most sensitive; positive in 70–80%
- IA-2A (islet antigen-2 antibodies) — in 60–70%
- ZnT8A (zinc transporter 8 antibodies) — in 60–70%
- IAA (insulin autoantibodies) — important before insulin therapy begins
C-peptide — marker of residual insulin secretion. Markedly reduced or undetectable in T1DM. Distinguishes T1DM (low C-peptide) from T2DM (normal or elevated) and LADA.
Blood glucose and HbA1c — current and medium-term glycaemic assessment.
Urine ketones and blood β-hydroxybutyrate — when ketoacidosis is suspected.
Ketoacidosis — The Primary Threat at Onset and Decompensation
Diabetic ketoacidosis (DKA) is the most life-threatening acute complication of T1DM — often the initial presentation in undiagnosed patients or the result of missed insulin injections.
Mechanism: absolute insulin deficiency → intense lipolysis → hepatic ketogenesis → accumulation of β-hydroxybutyrate and acetoacetate → metabolic acidosis.
DKA symptoms:
- Nausea, vomiting, abdominal pain
- Acetone breath ("fruity" odour)
- Kussmaul breathing — deep, rapid compensatory respiration
- Dehydration, dry skin and mucous membranes
- Impaired consciousness to coma
Laboratory findings: glucose > 11 mmol/L, urine ketones +++, pH < 7.3, bicarbonate < 15 mmol/L, potassium may be normal or elevated despite total body K⁺ depletion.
DKA treatment: inpatient only — fluid resuscitation, intravenous insulin, electrolyte correction. Mortality in specialist centres < 1%; without treatment, fatal.
Insulin Therapy and Glucose Monitoring in Type 1 Diabetes
T1DM requires lifelong insulin replacement therapy — this is not a choice but a biological necessity.
Insulin regimens
Basal-bolus regimen — the physiological standard:
- Basal insulin (long-acting: glargine, detemir, degludec) — once or twice daily, covering background requirements
- Bolus insulin (rapid-acting: aspart, lispro, glulisine) — before each meal, dose calculated by carbohydrate content
Insulin pump (CSII) — continuous subcutaneous infusion of rapid-acting insulin. More precisely mimics physiological secretion; indicated for unstable glycaemia and frequent hypoglycaemia.
Glucose monitoring
Traditional self-monitoring (glucometer) — minimum 4 measurements daily (before meals and before bed).
Continuous glucose monitoring (CGM) — a subcutaneous sensor measures glucose every 1–5 minutes. Has revolutionised glycaemic control: reduces hypoglycaemia frequency (especially nocturnal) and time outside target range. "Time in range" (TIR, 3.9–10 mmol/L) is more informative than HbA1c alone for assessing control quality.
HbA1c — every 3 months with unstable control; every 6 months when stable. Target for most T1DM patients: < 7.0%.
Hypoglycaemia — the primary treatment risk
Hypoglycaemia (glucose < 3.9 mmol/L) is the most frequent acute complication of insulin therapy.
Mild symptoms: tremor, sweating, palpitations, anxiety, hunger — adrenergic. Severe hypoglycaemia (< 2.8 mmol/L): confusion, seizures, loss of consciousness — requires external assistance.
Treatment: the "15–15 rule" — 15 g of fast-acting carbohydrates (3–4 glucose tablets, 150 mL juice), recheck after 15 minutes; repeat if symptomatic. If unconscious — intramuscular glucagon or intravenous 40% dextrose.
Closed-loop technology
"Artificial pancreas" systems automatically adjust the pump's insulin delivery based on real-time CGM data. Commercially available systems (Omnipod 5, MiniMed 780G) have demonstrated significant TIR improvement and reduced hypoglycaemia frequency.
Long-Term Complications of T1DM
Chronic hyperglycaemia damages blood vessels and nerves:
Microvascular: diabetic retinopathy (leading cause of adult blindness), nephropathy (progresses to CKD), neuropathy (painful, autonomic).
Macrovascular: accelerated atherosclerosis → myocardial infarction, stroke, peripheral artery disease.
Each 1% reduction in HbA1c reduces the risk of microvascular complications by 25–35% — a powerful rationale for tight glycaemic control.
When to Seek Urgent Medical Attention
- Thirst + frequent urination + weight loss in a child or young adult — rule out T1DM; check blood glucose immediately
- Acetone breath, vomiting, abdominal pain in a patient with diabetes — signs of DKA; call emergency services immediately
- Loss of consciousness in a patient with diabetes — hypoglycaemia or DKA; emergency services
- Glucose > 15 mmol/L + urine ketones — do not wait; go to emergency care
This article is for informational purposes only and does not replace consultation with a qualified endocrinologist.
Frequently Asked Questions
T1DM is an autoimmune disease with absolute insulin deficiency: the pancreas produces no insulin at all. It typically begins in childhood or young adulthood; insulin is required from day one. T2DM is insulin resistance with relative insulin deficiency: the pancreas produces insulin, but cells do not respond to it. It begins more often after age 40, is linked to obesity, and initial treatment is lifestyle change and oral medication.
Not currently. Destroyed β-cells do not regenerate. Research in islet transplantation and gene therapy is ongoing. The closest approach to a cure today is pancreas or islet transplantation, but it requires lifelong immunosuppression and is not available to all patients. With insulin therapy combined with continuous glucose monitoring, a full and active life is achievable with proper management. Long-term monitoring includes regular kidney function testing to detect nephropathy early.
Diabetic ketoacidosis is a life-threatening acute complication of insulin deficiency. Without insulin, fat is broken down producing ketone bodies → the blood becomes acidic → all organ functions deteriorate. Signs: acetone breath, vomiting, abdominal pain, deep breathing, impaired consciousness. Without inpatient treatment, it is fatal. At the first signs — call emergency services immediately.
A minimum of 4 times daily (before each meal and before bed). Optimally — continuous glucose monitoring (CGM), providing readings every 1–5 minutes with alerts for hypo- and hyperglycaemia. HbA1c is checked every 3 months with unstable control and every 6 months once targets are achieved.
With symptoms of hypoglycaemia (tremor, sweating, palpitations) and glucose < 3.9 mmol/L — immediately take 15 g of fast-acting carbohydrates: 3–4 glucose tablets, 150 mL juice or a sugary drink. Recheck glucose after 15 minutes — if not normalised, repeat. If the patient loses consciousness — administer intramuscular glucagon (every T1DM patient should have it) and call emergency services.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service