Uric Acid in Blood: Normal Levels, High Causes and Gout
A sudden, excruciating attack of pain in the big toe in the middle of the night — so intense that the weight of a sheet is unbearable — is the classic first presentation of gout. But by the time that pain finally announces itself, uric acid has been silently accumulating in the blood for years, crystallising in joints and damaging renal tubules. Uric acid is the final breakdown product of purines in the body. Its blood level reflects diet, kidney function, the rate of cellular turnover, and a range of metabolic disorders simultaneously. This article covers normal ranges for every group, causes of deviation, and what to do when the level is chronically elevated.
What Is Uric Acid and Why Is It Tested?
Purines are nitrogen-containing compounds that form the building blocks of DNA and RNA in every cell. When cells renew, age, or break down, purines are metabolised into uric acid. It is produced in the liver and cleared primarily by the kidneys — approximately 70% of the daily load — with the remainder excreted via the gut. Under normal conditions, production and excretion are in equilibrium. When this balance is disrupted — through excess production or insufficient excretion — uric acid accumulates in the plasma.
At physiological concentrations, uric acid is not a villain: it has antioxidant properties and protects cells from oxidative damage. The problem begins when its concentration chronically exceeds the solubility threshold — approximately 360 µmol/L for women and 420 µmol/L for men. Above these levels, uric acid crystallises as monosodium urate and deposits in tissues.
A uric acid test is ordered in several situations:
- Diagnosis and monitoring of gout
- Assessment of urinary stone risk (urate kidney stones)
- Chronic kidney disease — uric acid both damages kidneys and accumulates when they are impaired
- Metabolic syndrome, obesity, type 2 diabetes
- Monitoring during chemotherapy — tumour cell breakdown sharply elevates the level
- Long-term use of diuretics — particularly thiazides
For a comprehensive evaluation of renal excretory function, uric acid is assessed alongside the kidney function test: declining eGFR and rising uric acid frequently travel together.
How to Prepare and Get Tested
Uric acid is sensitive to diet and lifestyle — preparation errors produce false results that mislead both patient and doctor.
Preparation guidelines:
- Blood is drawn fasting in the morning — at least 8–12 hours after the last meal.
- Avoid alcohol for 24 hours: even a small amount of beer or wine inhibits renal urate excretion and transiently raises levels by 20–30%.
- Limit purine-rich foods for 24 hours: red meat, organ meats, seafood, anchovies, sardines, and legumes. A large meat portion the day before can produce a false elevation.
- Avoid intense exercise for 24 hours: increased muscle cell breakdown raises uric acid.
- Tell your doctor about all medications: low-dose aspirin, thiazide diuretics, cyclosporin, and pyrazinamide raise uric acid; allopurinol, fenofibrate, and high-dose aspirin lower it.
- For monitoring gout therapy or allopurinol treatment — always test at the same time of day and in the same laboratory.
A 24-hour urine uric acid test may also be ordered: it distinguishes overproduction (> 700 mg/day in urine) from underexcretion (< 700 mg/day despite high serum levels) — a crucial distinction for choosing treatment.
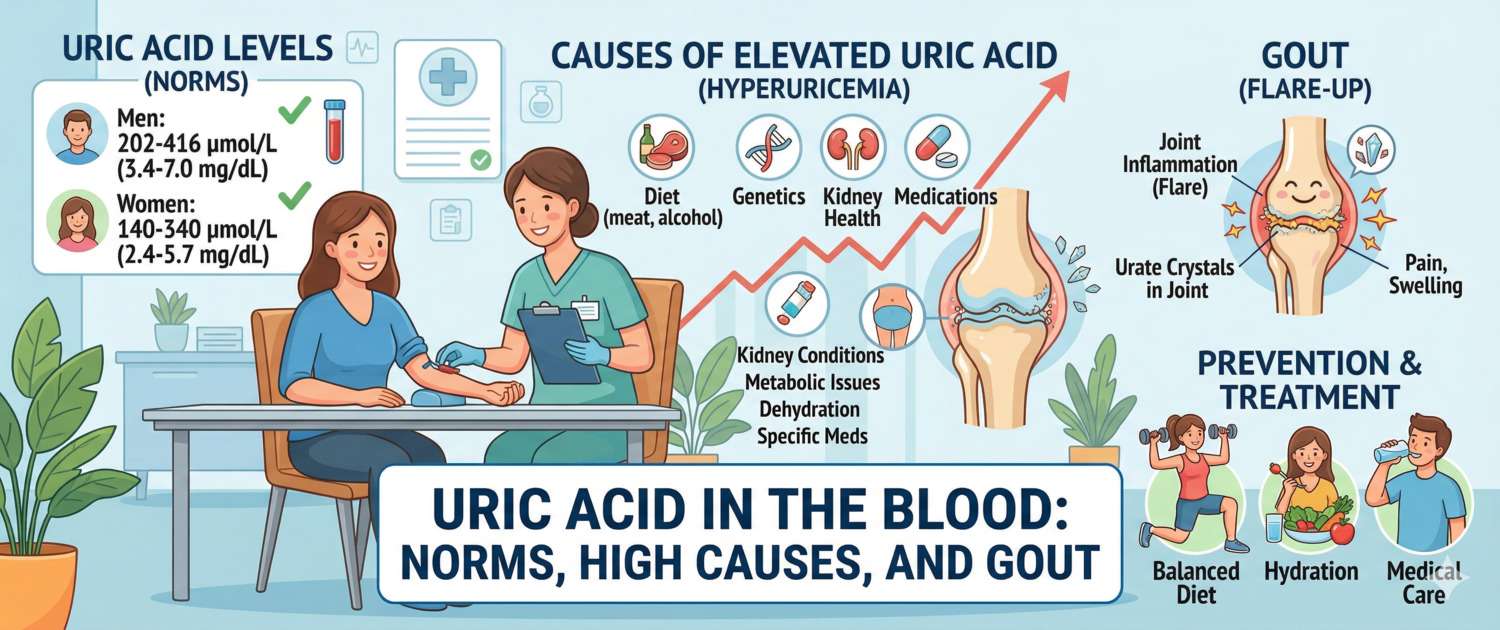
Uric Acid Normal Range: Table by Sex and Age
Normal values depend significantly on sex, age, and analytical method — always check the reference range on your own report.
| Category | Normal (µmol/L) | Normal (mg/dL) |
|---|---|---|
| Men 18–59 years | 200–430 | 3.4–7.2 |
| Men over 60 | 250–480 | 4.2–8.0 |
| Women 18–49 years | 140–360 | 2.4–6.0 |
| Women post-menopause | 200–430 | 3.4–7.2 |
| Children 1–12 years | 120–320 | 2.0–5.4 |
| Pregnant women | 120–270 | 2.0–4.5 |
In women of reproductive age, uric acid is substantially lower than in men — oestrogens enhance renal urate excretion. After menopause, this protective effect disappears, and normal values in older women approach those of men. This explains why gout in women is almost exclusively a post-menopausal disease.
The crystallisation threshold — 360 µmol/L for women and 420 µmol/L for men — is critically important for interpretation: values above these points create conditions for tissue crystal deposition even in the absence of obvious symptoms.
High Uric Acid (Hyperuricaemia): Causes and What It Means
Hyperuricaemia is defined as uric acid above 360 µmol/L in women or above 420 µmol/L in men. Three mechanisms account for the excess:
Overproduction — synthesis exceeds the kidney's capacity to excrete:
- Diet high in purines: red meat, organ meats, shellfish, alcohol (especially beer, rich in guanosine)
- Haematological malignancies with high cell turnover: leukaemia, lymphoma
- Tumour lysis syndrome during chemotherapy — massive cell breakdown
- Haemolytic anaemias, psoriasis, rhabdomyolysis
Underexcretion — kidneys fail to clear urates adequately:
- Chronic kidney disease — the leading cause of persistent hyperuricaemia in older adults
- Thiazide and loop diuretics — compete with urates for tubular secretion
- Dehydration — reduced renal blood flow concentrates urates
- Arterial hypertension
- Hypothyroidism — slows renal urate excretion
Mixed mechanism:
- Alcohol — simultaneously increases synthesis and inhibits excretion
- Insulin resistance and metabolic syndrome — insulin reduces renal uric acid clearance
- Obesity — elevated cellular turnover combined with reduced renal clearance
Asymptomatic hyperuricaemia — elevated uric acid without clinical manifestations — affects 5–8% of the population. Long considered benign, it is now understood to be an independent risk factor for hypertension, cardiovascular disease, and chronic kidney disease progression.
Gout: How Excess Urate Destroys Joints and Tissues
Gout is a chronic disease in which monosodium urate crystals deposit in joints, tendons, and soft tissues. Far from a simple "disease of excess," it is an immunoinflammatory condition with a precise pathogenesis.
Mechanism of an acute attack: urate crystals in the joint space are engulfed by neutrophils. The neutrophil attempts to digest the crystal — and destroys itself in the process, releasing a cascade of pro-inflammatory mediators. The result is acute inflammation with intense swelling, redness, heat, and unbearable pain. The classic site of the first attack is the first metatarsophalangeal joint — the base of the big toe.
Triggers of an acute attack: a heavy meal of meat and alcohol, dehydration, physical trauma, surgery, or — paradoxically — the start of allopurinol therapy, when a rapid fall in uric acid mobilises crystals from tissue deposits into the joint space.
In untreated chronic gout, tophi develop — firm, chalky nodules of urate crystals depositing under the skin over the ears, joints, and tendons. Tophi erode articular surfaces and may ulcerate.
A key clinical principle: gout is diagnosed clinically and confirmed by polarised light microscopy of synovial fluid — not by serum uric acid alone. During an acute flare, serum levels may be normal or even low, as crystals have migrated from plasma into the inflamed joint. Uric acid for diagnostic and therapeutic purposes is measured two to four weeks after the flare has resolved.
Uric Acid and the Kidneys: Urate Nephropathy and Kidney Stones
The kidneys are both the victim and the perpetuator of high uric acid. This bidirectional relationship makes the kidney central to managing patients with hyperuricaemia.
Urate nephropathy — chronic damage to the renal tubules and interstitium from urate crystal deposition. Mechanism: crystals accumulate in the renal medulla, triggering inflammation and fibrosis. Clinically: slowly progressive decline in kidney function, often with mild proteinuria. A rising creatinine trend in a patient with chronic hyperuricaemia should raise suspicion of urate-related renal injury.
Acute uric acid nephropathy — massive tubular obstruction by urate crystals, causing acute obstructive renal failure. The classic scenario is tumour lysis syndrome during chemotherapy. Uric acid exceeds 700–900 µmol/L, and urine turns dark brown. This is a medical emergency requiring immediate hydration and rasburicase administration.
Urate nephrolithiasis — kidney stone disease with uric acid stones. Accounts for approximately 10% of all renal stones. A distinctive feature: urate stones are radiolucent — invisible on plain X-ray, but clearly visible on ultrasound and CT. Risk factors: acidic urine (pH < 5.5), dehydration, high purine intake. Unlike calcium stones, urate stones can be dissolved medically by alkalinising the urine. The mineral workup in recurrent stone disease always includes calcium and phosphorus alongside uric acid.
An important therapeutic point: lowering uric acid with allopurinol in chronic kidney disease slows its progression — this has been demonstrated in several randomised controlled trials.
Low Uric Acid (Hypouricaemia): When Does It Matter?
Hypouricaemia — uric acid below 120 µmol/L in adults — is far less common than hyperuricaemia and carries less clinical weight, but several underlying causes warrant exclusion.
Causes of hypouricaemia:
- Fanconi syndrome and tubular reabsorption defects — the kidneys lose urates alongside other substances (glucose, amino acids, phosphorus)
- Extremely purine-poor diet — strict veganism with minimal protein intake
- Uricosuric agents — probenecid, losartan, high-dose aspirin
- Severe liver disease — impaired synthesis of xanthine oxidase, the enzyme that converts xanthine to uric acid
- Xanthinuria — a rare inherited disorder of purine metabolism
- SIADH — plasma dilution in syndrome of inappropriate ADH secretion
An isolated low uric acid in a patient on uricosuric therapy is an expected result, not a pathology. Persistently low levels without a clear cause warrant evaluation by a nephrologist or endocrinologist.
When Uric Acid Results Require Medical Attention
Mild asymptomatic hyperuricaemia without organ damage calls for dietary adjustment and a repeat test in three to six months. But several situations require prompt consultation:
- Uric acid above 540 µmol/L (9 mg/dL) — crystal deposition risk rises sharply
- A first acute arthritis flare with joint redness and pain — see a rheumatologist promptly
- Uric acid above 700 µmol/L in a patient with a malignancy or after chemotherapy — risk of acute urate nephropathy
- Persistent elevation combined with rising creatinine — possible urate kidney injury
- Discovery of tophi or radiologically confirmed joint damage
- Recurrent kidney stones of the urate type
- Uric acid above 480 µmol/L in a patient with hypertension or metabolic syndrome — discuss pharmacological lowering with your doctor
The treatment target in gout is below 360 µmol/L (6 mg/dL); with tophi, below 300 µmol/L. Reaching the target usually requires several months of therapy and regular monitoring.
Conclusion
Uric acid sits at the intersection of diet, kidney function, and purine metabolism. Its chronic elevation sets off a chain of organ damage — joints, kidneys, blood vessels — that develops silently over years. Normal ranges differ substantially by sex and age: in young women they are roughly half those of men the same age, equalising only after menopause. For mild asymptomatic hyperuricaemia, the first step is dietary and hydration changes. At levels above 480–540 µmol/L or with clinical manifestations, a rheumatology or nephrology consultation and a discussion of pharmacological treatment are warranted.
For male-specific reference ranges, the link with metabolic syndrome and cardiovascular risk, see the article uric acid in men.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
For men aged 18–59, the normal range is 200–430 µmol/L (3.4–7.2 mg/dL); for men over 60, up to 480 µmol/L. For women of reproductive age, 140–360 µmol/L (2.4–6.0 mg/dL) — oestrogens enhance renal urate excretion. After menopause this protective effect is lost and normal values in older women approach those of men. The crystallisation threshold — above 360 µmol/L in women and 420 µmol/L in men — matters as much as the reference range: values above these points create conditions for urate crystal deposition regardless of whether symptoms are present.
Red meat, organ meats, and seafood are rich in purines — nitrogen-containing compounds that break down into uric acid. A large meat portion raises the level within hours. Alcohol acts by a double mechanism: beer contains guanosine, a direct uric acid precursor, while ethanol inhibits renal urate excretion by competing for tubular transport. This is why a heavy meal of meat combined with alcohol is the classic trigger of an acute gout flare.
Yes — and this is a common diagnostic trap. During an acute flare, urates actively migrate from the plasma into the inflamed joint, and the serum level may normalise or even fall below normal. Gout is confirmed by polarised light microscopy of synovial fluid or clinically, not by serum uric acid alone. For diagnostic and therapeutic purposes, uric acid is measured two to four weeks after the flare has resolved, not during the acute episode.
It was long considered a harmless laboratory finding, but current evidence disagrees: chronically elevated uric acid is an independent risk factor for hypertension, cardiovascular events, and kidney disease progression. It almost always accompanies metabolic syndrome — patients with high uric acid frequently also have elevated triglycerides, insulin resistance, and high blood pressure. This makes a comprehensive metabolic workup important, not just isolated monitoring of uric acid.
Dietary changes are effective for mild hyperuricaemia: limit red meat, organ meats, shellfish, and beer; increase daily water intake to 2–2.5 litres (diluting urates and enhancing renal excretion); lose weight if obese (each 5 kg lost reduces uric acid by approximately 30–40 µmol/L); avoid large amounts of fructose — sugary drinks and juice increase urate synthesis. Dairy products, conversely, have a mild urate-lowering effect. However, at levels above 480–540 µmol/L or with established gout, diet alone is insufficient.
Yes — the relationship is bidirectional. Insulin resistance — the central mechanism of type 2 diabetes — reduces renal uric acid clearance, so hyperuricaemia frequently precedes the clinical onset of diabetes. Conversely, high uric acid amplifies oxidative stress in the pancreas and worsens insulin sensitivity. In type 2 diabetes, uric acid is recommended for annual monitoring — its chronic elevation accelerates diabetic nephropathy and should be treated alongside blood glucose and blood pressure.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service