Hepatocellular Carcinoma: Symptoms, Diagnosis and Treatment
Most patients with hepatocellular carcinoma already have serious liver disease — cirrhosis, chronic hepatitis or fatty liver — long before the tumour makes itself known. This is why liver cancer ranks fifth among cancer deaths worldwide despite a comparatively modest share of overall incidence: it is almost always found late, in a patient who is already seriously ill.
What Hepatocellular Carcinoma Is
Hepatocellular carcinoma (HCC) is a malignant tumour arising from hepatocytes — the principal cells of the liver parenchyma. It accounts for 90% of all primary liver malignancies. It must not be confused with metastatic liver cancer, which occurs 20–30 times more frequently (breast, colorectal, gastric and other cancers regularly spread to the liver) and requires an entirely different approach to treatment.
80–90% of HCC develops on a background of liver cirrhosis. This is not coincidental: chronic inflammation, fibrosis and accelerated hepatocyte turnover during regeneration create the ideal environment for oncogenic mutation accumulation. The path from a healthy liver to HCC takes years to decades — typically passing through chronic hepatitis or progressive fibrosis.
Approximately 900,000 new cases of HCC are diagnosed worldwide each year. Incidence continues to rise, driven predominantly by the epidemic of non-alcoholic fatty liver disease (NAFLD) and its metabolic sequela, cirrhosis. While viral hepatitis B and C can be treated and controlled, the wave of NAFLD-associated HCC is still growing.
BCLC (Barcelona Clinic Liver Cancer) staging incorporates not only tumour size but also liver function according to the Child–Pugh score and the patient's performance status — this integrated approach determines treatment strategy:
| BCLC stage | Description | Recommended treatment |
|---|---|---|
| 0 (very early) | Single tumour < 2 cm, preserved liver function | Resection or ablation |
| A (early) | 1–3 tumours ≤ 3 cm, compensated cirrhosis | Resection, transplant, ablation |
| B (intermediate) | Multinodular, no vascular invasion | TACE |
| C (advanced) | Vascular invasion or metastases | Systemic therapy |
| D (terminal) | Decompensated cirrhosis | Palliative care |
Liver Cancer Risk Factors: Cirrhosis, Hepatitis and Beyond
Chronic hepatitis B virus (HBV) — the leading cause of HCC globally, responsible for 50–55% of all cases. HBV integrates into the hepatocyte genome and directly activates oncogenes even before cirrhosis develops — distinguishing it from most other causes, where HCC arises almost exclusively in cirrhotic tissue. HBV vaccination is the only proven oncological prevention measure, reducing HCC incidence by 60–70% in vaccinated populations.
Chronic hepatitis C virus (HCV) — accounts for 25–30% of cases in high-income countries. Unlike HBV, HCV causes HCC almost exclusively through cirrhosis. Modern direct-acting antivirals (DAAs) achieve viral clearance in 95–99% of patients, but the risk of HCC in patients who already have cirrhosis persists for years after sustained virological response — these patients must continue surveillance.
Chronic hepatitis and cirrhosis from any cause: alcohol, autoimmune hepatitis, primary biliary cholangitis, Wilson's disease or hereditary haemochromatosis.
Fatty liver disease (NAFLD) — a rapidly growing cause of HCC in Western countries. A clinically important feature: HCC in NAFLD develops before cirrhosis in approximately 20–30% of cases — substantially complicating screening. Obesity and type 2 diabetes, the main drivers of NAFLD, are simultaneously independent risk factors for HCC.
Alcohol raises HCC risk two- to threefold; when combined with hepatitis B or C, risks multiply.
Aflatoxin B1 — a mycotoxin produced in mouldy groundnuts, cereals and maize in tropical climates. A direct mutagen targeting the TP53 tumour-suppressor gene. In combination with HBV infection, risk of HCC increases approximately 60-fold.
Genetic conditions: haemochromatosis (iron overload), Wilson's disease (copper accumulation) and alpha-1-antitrypsin deficiency — all three impose chronic toxic stress on hepatocytes and lead to cirrhosis with elevated HCC risk.
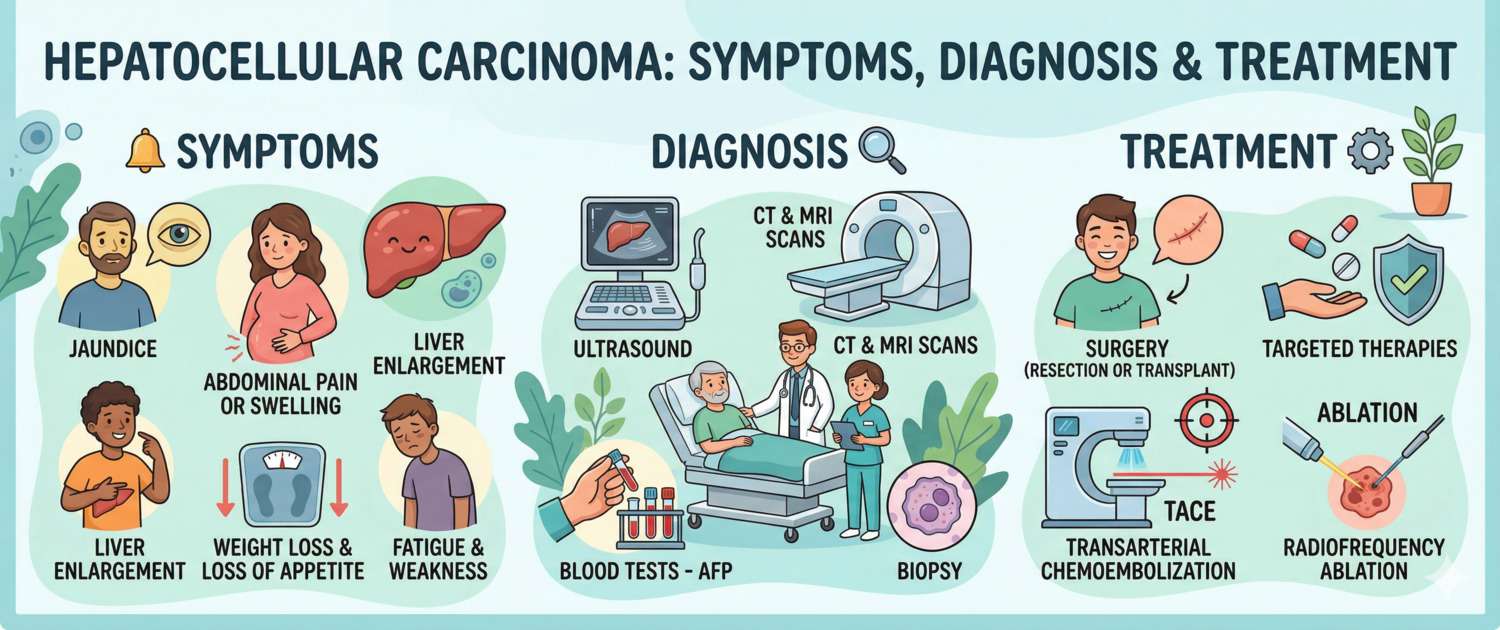
Early Signs and Symptoms of Liver Cancer: Why HCC Is Diagnosed Late
The liver parenchyma contains no pain receptors. A tumour can grow to a considerable size without causing pain or visible functional impairment. Early symptoms often blend imperceptibly with manifestations of underlying cirrhosis or chronic hepatitis.
Symptoms that warrant investigation:
- New or worsening discomfort in the right upper abdomen — in a patient with cirrhosis this is an important signal even when attributed to "usual" sensations
- Sudden deterioration of previously compensated cirrhosis: worsening ascites, new encephalopathy or jaundice without an obvious precipitating cause
- Unexplained weight loss and progressive weakness
- A palpable mass in the right upper quadrant
Complications that often constitute the initial clinical presentation:
- Tumour rupture with haemoperitoneum — sudden severe abdominal pain, haemodynamic instability, requires emergency surgery
- Portal vein thrombosis — abrupt worsening of ascites and variceal haemorrhage
- Paraneoplastic syndromes: hypoglycaemia (IGF-2 secretion by the tumour), hypercalcaemia, erythrocytosis (ectopic erythropoietin production)
Jaundice in HCC is a late sign, indicating either bile duct compression by the tumour or decompensation of liver function. Its appearance in a previously compensated cirrhotic patient is an urgent signal requiring immediate investigation.
The majority of HCC cases detected today are found on surveillance ultrasound in patients with known cirrhosis — incidentally, not because of symptoms. This again underscores why regular screening in this group is critically important.
Diagnosis: Imaging, Biopsy and Laboratory Tests
HCC diagnosis is unique in that it does not always require biopsy: a characteristic pattern on contrast-enhanced imaging is considered sufficient for diagnosis in the appropriate clinical setting.
Dynamic contrast-enhanced CT or MRI — the gold standard. The diagnostic hallmark of HCC is "arterial phase hyperenhancement with washout": the tumour avidly enhances in the arterial phase (due to abundant neovascularisation) and rapidly loses contrast in the portal venous and delayed phases — unlike the surrounding liver tissue. This pattern for a nodule > 1 cm in a cirrhotic liver is pathognomonic for HCC: biopsy is not required.
Image-guided biopsy is indicated for atypical imaging appearances or in the absence of cirrhosis. The main risk is rare but documented needle-track tumour seeding.
Ultrasound — the first-line screening modality: twice yearly in all patients with cirrhosis. Any nodule > 1 cm detected on surveillance requires immediate CT or MRI confirmation.
Laboratory tests:
Liver function tests — assessment of functional reserve: bilirubin, albumin and prothrombin time constitute the Child–Pugh score that determines resectability. Rising bilirubin and falling albumin signal decompensation.
Complete blood count — thrombocytopaenia and leucopaenia from hypersplenism as markers of portal hypertension severity; normochromic anaemia in chronic disease.
Tumour markers — AFP and PIVKA-II (also known as des-gamma-carboxyprothrombin, DCP): both used for screening, diagnosis and treatment monitoring in HCC.
AFP Tumour Marker: Role in Diagnosis and Monitoring
AFP (alpha-fetoprotein) — the principal biomarker for HCC. Normal range in adults < 10–20 ng/mL (laboratory-dependent). Physiologically elevated AFP is present only in the fetus and in pregnant women.
Sensitivity and specificity in HCC: At a threshold of 20 ng/mL: sensitivity 60%, specificity 90%. At 200 ng/mL: sensitivity falls to ~22%, but specificity rises to 99%. A markedly elevated AFP (> 400–500 ng/mL) combined with characteristic CT/MRI findings is virtually diagnostic of HCC.
Limitations of AFP:
- 30–40% of patients with HCC have a normal AFP — particularly with small tumours and the fibrolamellar subtype. Normal AFP does not exclude cancer
- False-positive elevation occurs in chronic hepatitis flares, cirrhosis, acute liver failure and pregnancy
- Elevated AFP without a visible lesion on ultrasound in a hepatitis patient warrants repeat imaging at 3 months, not immediate biopsy
Clinical applications:
- Screening: AFP + ultrasound every 6 months in all cirrhotic patients
- Monitoring: AFP decline after surgery, ablation or systemic therapy is a marker of response; rising AFP signals progression or recurrence
- A baseline AFP > 1000 ng/mL is associated with more aggressive disease and is used as a selection criterion in several transplantation protocols
Treatment of Hepatocellular Carcinoma
Treatment of HCC faces a dual challenge: attacking the tumour while preserving what remains of an already-damaged cirrhotic liver. Treatment selection is guided by BCLC stage.
Surgical resection — the optimal approach at BCLC stage 0/A in the absence of cirrhosis or in compensated cirrhosis (Child–Pugh A) with sufficient residual parenchymal volume. Five-year survival: 50–70%. The primary limitation is that cirrhosis in most patients does not allow adequate resection without risk of post-operative liver failure.
Liver transplantation — a radical option that simultaneously removes the tumour and eliminates cirrhosis as the source of future malignancy. The Milan criteria (single tumour ≤ 5 cm or up to 3 tumours each ≤ 3 cm, no macrovascular invasion, no extrahepatic spread) define suitable candidates; five-year survival reaches 70–75%. Extended criteria (UCSF, "up-to-seven" rule) allow inclusion of more patients with comparable outcomes. The primary barrier is the shortage of donor organs and waiting list time.
Ablation — tumour destruction by physical methods: radiofrequency ablation (RFA) or microwave ablation. Effective for nodules ≤ 3 cm that are not amenable to resection. Five-year survival for small tumours is comparable to surgery — 50–60%. Minimally invasive; also used as a "bridge" to transplantation.
Transarterial chemoembolisation (TACE) — the standard for intermediate-stage BCLC B (multinodular HCC without vascular invasion). A catheter delivers embolic material combined with a chemotherapeutic agent (doxorubicin, cisplatin) into the hepatic artery feeding the tumour, causing ischaemia and local cytotoxic effect. Median survival: 20–26 months; also used as a bridge to transplantation.
Systemic therapy (BCLC C):
- Atezolizumab + bevacizumab — the first-line standard: a combination of anti-PD-L1 immunotherapy and anti-VEGF antiangiogenic therapy. In the IMbrave150 trial, median overall survival was 19.2 months versus 13.4 months with sorafenib monotherapy
- Sorafenib — the first targeted agent approved for HCC (2007); a multikinase inhibitor blocking angiogenesis and tumour proliferation; used when immunotherapy is contraindicated
- Lenvatinib — an alternative first-line option for patients without main portal vein invasion
- Second-line options: nivolumab, pembrolizumab, ramucirumab (for AFP ≥ 400 ng/mL)
Radiotherapy: stereotactic body radiotherapy (SBRT) is an emerging option for tumours not amenable to ablation or with tumour portal vein thrombus.
Screening and When to See a Doctor
HCC surveillance is strongly recommended and proven effective for high-risk groups. Abdominal ultrasound plus AFP every 6 months is the international standard of care for:
- all patients with cirrhosis from any cause (Child–Pugh A and B)
- patients with chronic hepatitis B even without cirrhosis — when viral load is high or there is a family history of HCC
- patients with chronic hepatitis C who have achieved sustained virological response but had established cirrhosis at the time of treatment
Regular surveillance reduces HCC mortality by 37–39% in cohort studies. Early detection through screening is the reason countries with strong surveillance programmes achieve significantly higher rates of curative treatment.
See a gastroenterologist or hepatologist without delay when any of the following apply:
- you have cirrhosis or chronic hepatitis and have not had surveillance ultrasound with AFP within the past 6 months;
- new or worsening pain in the right upper abdomen has developed;
- previously compensated cirrhosis has suddenly worsened — accumulating ascites, new jaundice;
- significant weight loss and weakness have developed on a background of chronic liver disease;
- AFP measured for any other reason has come back elevated.
HCC is substantially more treatable at early stages. Five-year survival at BCLC stage 0/A is 50–70%; at BCLC stage C it falls to below 15%. Complete HBV elimination through vaccination and HCV eradication with direct-acting antivirals are the most powerful preventive tools available. Do not delay either surveillance or treatment of underlying liver disease.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
In 80–90% of cases, yes. But there are important exceptions. With chronic hepatitis B, HCC can arise in a non-cirrhotic liver — the virus integrates into the hepatocyte genome and directly activates oncogenes. In fatty liver disease, HCC develops before cirrhosis in approximately 20–30% of cases, complicating surveillance. Fibrolamellar carcinoma — a rare HCC subtype in young patients — almost invariably arises in a non-cirrhotic liver. This is why surveillance for chronic hepatitis B patients begins regardless of whether cirrhosis is present.
AFP is used in three clinical contexts: screening in cirrhotic patients (combined with ultrasound every 6 months), monitoring treatment response, and early detection of recurrence after surgery or ablation. Key limitations: 30–40% of HCC patients have a normal AFP — particularly with small tumours. A concurrent rise in LDH alongside AFP elevation in a patient with worsening symptoms is an additional laboratory signal of disease progression that should prompt urgent imaging.
Yes — and transplantation is the only method that simultaneously eliminates both the tumour and cirrhosis as the source of future cancer. Suitable candidates are defined by the Milan criteria: a single tumour ≤ 5 cm or up to 3 tumours each ≤ 3 cm, no macrovascular invasion, no extrahepatic metastases. Five-year survival is 70–75%. The primary barrier is a shortage of donor organs: waiting list times are often months to years. During the wait, ablation or TACE are used to keep the tumour within transplant criteria.
Any chronic liver disease progressing to cirrhosis: chronic hepatitis B and C, alcoholic cirrhosis, non-alcoholic steatohepatitis (the fastest-growing cause), primary biliary cholangitis, hereditary haemochromatosis, Wilson's disease and alpha-1-antitrypsin deficiency. Autoimmune hepatitis diagnosed late or inadequately treated progresses to cirrhosis with HCC risk comparable to viral hepatitis. All patients with these conditions should be enrolled in a regular surveillance programme.
Conventional intravenous chemotherapy is poorly effective in HCC — the cirrhotic liver tolerates its toxicity badly and hepatocytes possess robust drug resistance mechanisms. Locoregional chemotherapy delivered via TACE is a different matter: the drug is targeted directly into the tumour through its feeding artery. For systemic disease, the current standard is immunotherapy (atezolizumab + bevacizumab) and targeted agents (sorafenib, lenvatinib) — not classical cytotoxic chemotherapy.
Every 6 months together with abdominal ultrasound — this is the international surveillance standard for all patients with cirrhosis, regardless of its cause. Ultrasound alone without AFP reduces the sensitivity of screening: their combined use raises early-stage detection rates from approximately 45% to 63%. A single normal AFP does not exclude cancer — the trend over time matters. A rapid rise in AFP even within the normal range (for example, from 5 to 18 ng/mL over 3 months) warrants an unscheduled ultrasound and hepatology review, as this dynamic can precede a visible lesion on imaging by several months.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service