Adrenal Insufficiency: Symptoms, Diagnosis and Treatment
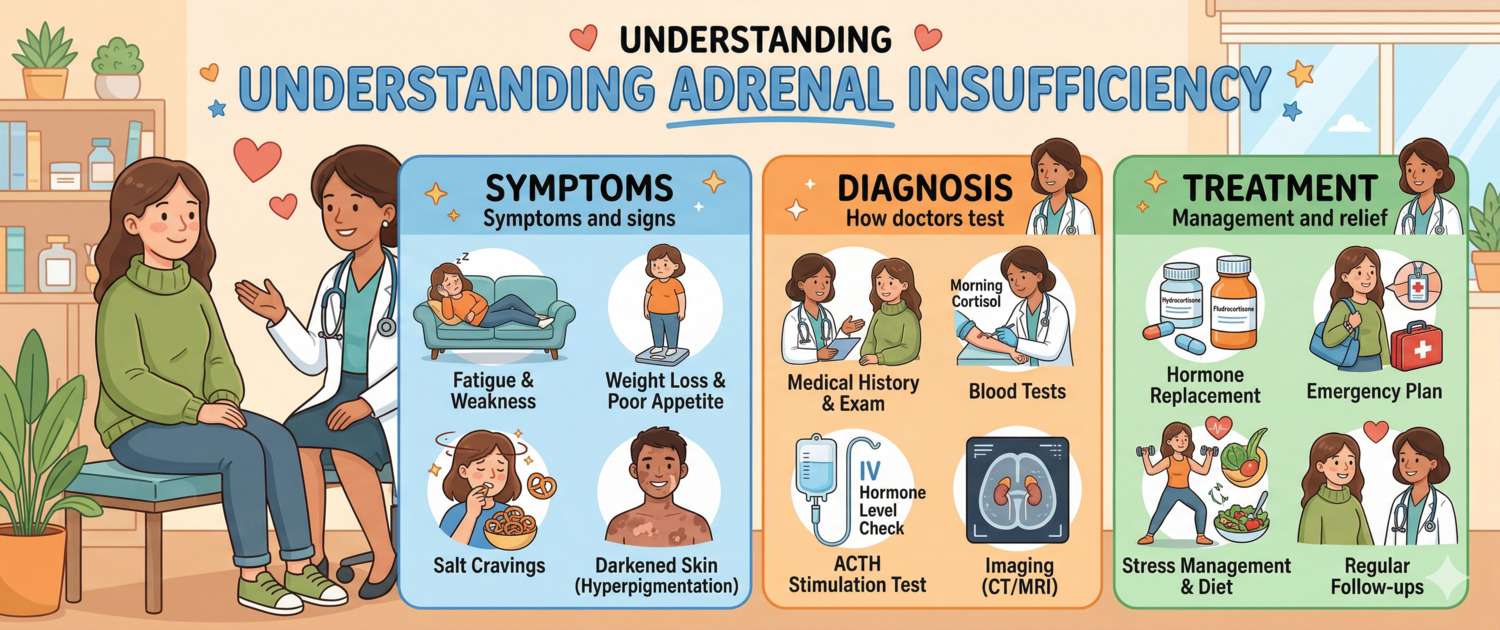
Fatigue that never improves with rest, unexplained weight loss, a craving for salty food, and skin that darkens in places the sun never reaches — this rare combination of symptoms should immediately prompt a physician to consider adrenal insufficiency. The adrenal glands produce hormones without which the body cannot respond normally to stress. When this function is impaired, even a routine cold can become a life-threatening crisis.
What Adrenal Insufficiency Is
Adrenal insufficiency (AI) is a condition in which the adrenal cortex produces inadequate amounts of steroid hormones — primarily cortisol and aldosterone. Cortisol — the "stress hormone" — regulates metabolism, immune response, and the body's reaction to physiological demands. Aldosterone controls the balance of sodium and potassium, maintaining blood pressure.
Think of the body as an emergency service: under any threat — infection, trauma, surgery — cortisol is normally released into the bloodstream and mobilises resources. In AI, this mobilisation fails, and the body cannot cope even with moderate stress. This is why AI is sometimes called "the disease that kills in stressful situations."
Causes: Primary and Secondary Forms
The key distinction is where the problem originates.
Primary adrenal insufficiency (Addison's disease) — damage to the adrenal glands themselves. The cortex is destroyed and both hormones — cortisol and aldosterone — are no longer produced in adequate amounts. Causes in order of frequency: autoimmune adrenalitis (80–90% of cases in developed countries) — autoantibodies attack the adrenal cortex; adrenal tuberculosis — historically the leading cause, now rare; adrenal metastases; bilateral adrenalectomy; adrenal haemorrhage (Waterhouse-Friderichsen syndrome in sepsis).
Autoimmune AI frequently co-occurs with other autoimmune diseases — Hashimoto's thyroiditis and type 1 diabetes. This combination is called autoimmune polyendocrine syndrome.
Secondary adrenal insufficiency — the adrenal glands are intact, but the pituitary gland does not produce enough ACTH (adrenocorticotrophic hormone) to stimulate them. Causes: pituitary tumours, surgery or irradiation in that region, Sheehan's syndrome (pituitary ischaemia after postpartum haemorrhage). The most important cause is abrupt withdrawal of long-term glucocorticoid therapy: chronic prednisolone use suppresses the HPA axis, which recovers slowly. Sudden discontinuation is the classic path to acute crisis.
Key distinction: in primary AI, aldosterone is absent → hyponatraemia + hyperkalaemia. In secondary AI, aldosterone is preserved (regulated through a separate pathway) → electrolyte disturbances are less pronounced.
Symptoms of Adrenal Insufficiency
Chronic AI develops gradually — over months to years. Symptoms are non-specific and are frequently attributed to depression, chronic fatigue syndrome, or anorexia.
General symptoms: progressive weakness and fatigue — the earliest and most prominent symptom; weight loss and poor appetite; nausea, vomiting, and abdominal pain; muscle aches and cramps; orthostatic hypotension — dizziness on standing.
Specific to primary AI: hyperpigmentation of skin and mucous membranes — darkening of palmar creases, surgical scars, areolae, and the inner cheeks. The mechanism: excess ACTH (the pituitary "shouting" at the adrenals) cross-stimulates melanocytes. Salt craving — the body attempts to compensate for sodium loss. Low blood pressure, potentially progressing to collapse.
Symptoms of cortisol deficiency: hypoglycaemia (especially with fasting or exercise), impaired stress tolerance, cognitive fog, depression.
Diagnosis: Key Tests
Diagnosing AI is a multi-step process. No single test establishes the diagnosis on its own.
Morning cortisol (drawn 8:00–9:00 AM) — the first step. Cortisol is highest in the morning: a value < 140 nmol/L strongly suggests AI; > 500 nmol/L effectively excludes it. The "grey zone" of 140–500 nmol/L requires stimulation testing.
Synacthen stimulation test (250 µg ACTH) — the gold standard. Synthetic ACTH is injected and cortisol is measured at 30 and 60 minutes. A normal response: peak > 550 nmol/L. Failure to mount an adequate response confirms adrenal insufficiency.
Plasma ACTH — differentiates primary from secondary AI. In primary AI, ACTH is markedly elevated (the pituitary "shouting"). In secondary AI, ACTH is low or normal.
Sodium and potassium — in primary AI, hyponatraemia (< 135 mmol/L) and hyperkalaemia (> 5.0 mmol/L) are characteristic. Assessed comprehensively via the electrolyte panel. This combination alongside fatigue and hypotension is a strong pointer to primary AI. The mirror pattern — low potassium with normal or elevated sodium and resistant hypertension — is characteristic of primary aldosteronism and requires an entirely different management approach.
Fasting glucose — hypoglycaemia in AI reflects cortisol's role in maintaining blood sugar through gluconeogenesis.
Anti-adrenal antibodies (anti-21-hydroxylase) — confirm the autoimmune aetiology in primary AI.
Pituitary MRI — in secondary AI to exclude an adenoma or other structural pathology.
Adrenal Crisis: When to Call Emergency Services
Acute adrenal insufficiency (adrenal crisis) is a life-threatening emergency. It develops when a patient with AI faces severe infection, trauma, or surgery without an adequate increase in their hormone dose, or when glucocorticoids are abruptly withdrawn.
Signs of crisis: sudden profound weakness preventing the patient from getting up; severe abdominal pain with nausea and vomiting; critical blood pressure drop (systolic < 90 mmHg); confusion or loss of consciousness; hypoglycaemia.
If adrenal crisis is suspected, call emergency services immediately. Treatment: intravenous hydrocortisone 100 mg bolus + aggressive normal saline infusion. Delaying hormone administration to draw blood tests is never appropriate — the mortality risk is high.
Treatment of Adrenal Insufficiency
Treatment of AI requires lifelong hormone replacement therapy. The goal is to replicate the normal diurnal cortisol rhythm.
Glucocorticoids: hydrocortisone 15–25 mg/day in 2–3 doses (larger morning dose, smaller afternoon dose, none in the evening). Or prednisolone 5–7.5 mg/day. The "sick day rule": during fever, vomiting, or surgery, the dose is doubled or tripled. If oral administration is impossible, the dose is given parenterally.
Mineralocorticoids (primary AI only): fludrocortisone 0.05–0.2 mg/day — replaces aldosterone deficiency, normalising blood pressure and electrolytes.
Patient education — a critical component: every patient must carry a medical alert card, understand the sick day rule, and have an emergency kit with injectable hydrocortisone for crisis situations.
In autoimmune AI, the underlying process cannot be reversed — adrenal tissue is permanently destroyed. Treatment aims solely at hormone replacement.
When to Seek Urgent Medical Attention
Call emergency services immediately for: a sudden blood pressure drop, loss of consciousness, or confusion; uncontrolled vomiting that prevents taking oral medication; acute abdominal pain in a known AI patient; high fever in an AI patient who cannot increase their steroid dose.
See an endocrinologist within days for: progressive fatigue and falling blood pressure without obvious cause; unexplained skin hyperpigmentation; hyponatraemia combined with hyperkalaemia on blood tests.
This article is for informational purposes only. Diagnosis and treatment of adrenal insufficiency are carried out by an endocrinologist.
Frequently Asked Questions
Addison's disease is primary adrenal insufficiency, in which the adrenal cortex is destroyed — most often by an autoimmune process — and stops producing adequate cortisol and aldosterone. A hallmark feature is skin hyperpigmentation: unlike secondary AI, in Addison's disease the pituitary overproduces ACTH, which cross-stimulates melanocytes. Lifelong replacement with both glucocorticoids and mineralocorticoids is required.
The first step is a morning cortisol blood test (drawn at 8:00–9:00 AM): a value below 140 nmol/L strongly suggests AI. Confirmation requires a synacthen stimulation test. Simultaneously, an electrolyte panel is ordered — hyponatraemia combined with hyperkalaemia is the characteristic biochemical fingerprint of primary AI. Plasma ACTH and anti-21-hydroxylase antibodies help identify the cause and form.
In primary AI (Addison's disease), the adrenal glands are destroyed — both cortisol and aldosterone are deficient. This causes hyponatraemia, hyperkalaemia, and skin hyperpigmentation. ACTH is markedly elevated. In secondary AI, the adrenal glands are intact but the pituitary fails to send the ACTH signal: only cortisol is deficient, as aldosterone is regulated by a separate pathway. Hyperpigmentation is absent and electrolyte disturbances are milder. The most common cause of secondary AI is abrupt withdrawal after long-term glucocorticoid therapy.
An adrenal crisis is an acute, life-threatening collapse of adrenal function that occurs when a patient with AI faces a physiological stressor — infection, trauma, or surgery — without increasing their steroid dose. It presents as a catastrophic drop in blood pressure, vomiting, severe abdominal pain, and loss of consciousness. Treatment is emergency intravenous hydrocortisone 100 mg plus saline infusion. Delaying the hormone to draw blood tests is never justified — this is a true emergency.
Yes — with appropriate replacement therapy and proper patient education, most people with AI lead full, active lives. The key rules are: take steroids every day without skipping; double or triple the dose during illness or stress; always carry an emergency injectable hydrocortisone kit and a medical alert card. Regular endocrinology follow-up every 6–12 months is recommended.
Aldosterone — a hormone of the adrenal cortex — controls potassium handling in the kidneys: it drives sodium retention and potassium excretion. In primary AI, aldosterone is absent, so the kidneys stop excreting potassium → hyperkalaemia develops. Simultaneously, sodium is lost → hyponatraemia. This combination — hyponatraemia plus hyperkalaemia — is the biochemical hallmark of primary adrenal insufficiency specifically.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service