Antiphospholipid Syndrome: Symptoms, Diagnosis and Treatment
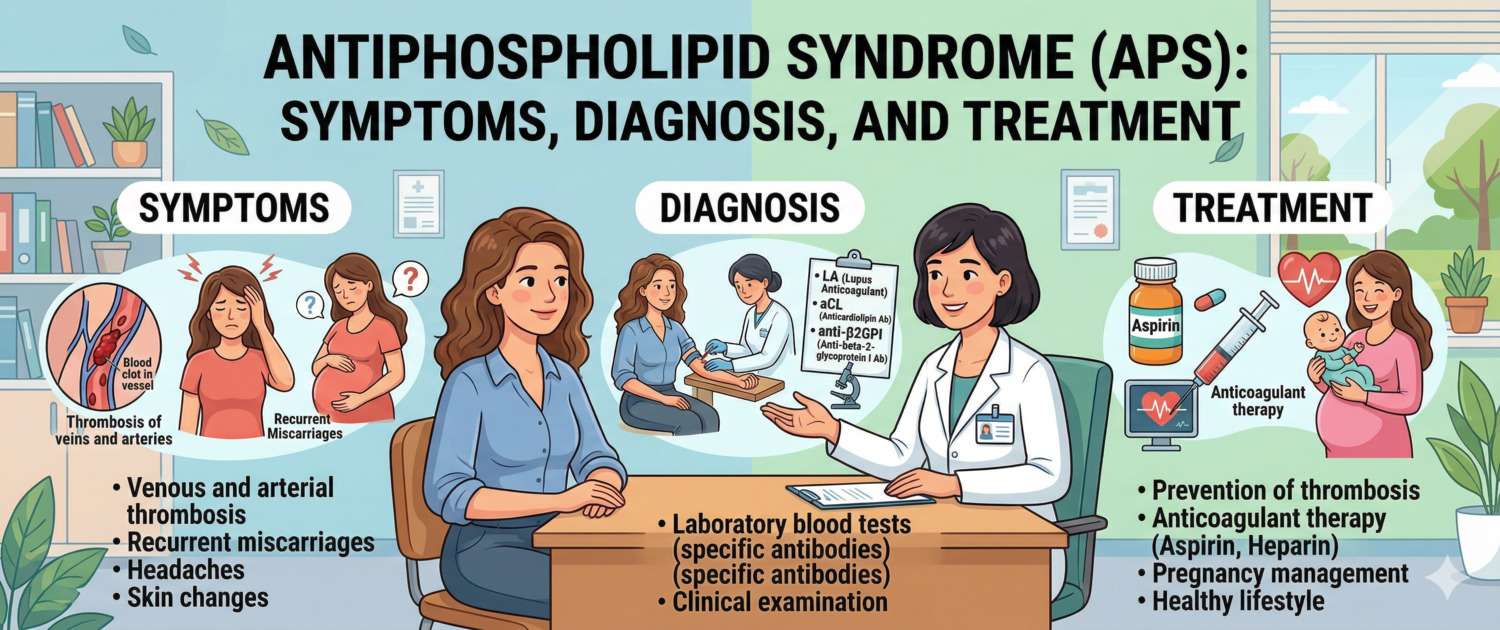
A young woman suffers a stroke without atherosclerosis or hypertension. Another has three consecutive miscarriages with no gynaecological explanation. A third patient — deep vein thrombosis at 30 with no apparent cause. Behind all three presentations may lie a single diagnosis: antiphospholipid syndrome. This autoimmune disease causes the immune system to produce antibodies that attack the body's own phospholipids, triggering pathological clot formation.
What Is Antiphospholipid Syndrome and How Does It Develop
Antiphospholipid syndrome (APS, Hughes syndrome) is an acquired autoimmune thrombophilic condition in which antiphospholipid antibodies (aPL) circulate in the blood, increasing the tendency to thrombosis in arteries and veins, and in pregnant women — to placental pathology and pregnancy loss.
In health, phospholipids — components of cell membranes — are inaccessible to the immune system. In APS, for reasons not fully understood, the immune system begins producing antibodies against phospholipids (primarily cardiolipin and beta-2-glycoprotein I) and their associated proteins. These antibodies disrupt the normal coagulation system at multiple points: they activate platelets and vascular endothelial cells, suppress natural anticoagulant mechanisms and impair fibrinolysis. The result is pathological clot formation in vessels of any calibre and any location.
APS belongs to the group of thrombophilias — conditions with an elevated tendency to clot. Its defining distinction from hereditary thrombophilias (protein C deficiency, factor V Leiden mutation) is its acquired and autoimmune nature. This means that unlike genetic defects, APS is theoretically amenable to treatment directed at the immune system.
APS may exist as a standalone condition (primary APS) or in the context of another autoimmune disease — most commonly systemic lupus erythematosus (secondary APS). Approximately 30–40% of patients with SLE have antiphospholipid antibodies, and a subset develops clinically overt APS.
Causes and Risk Factors for Antiphospholipid Syndrome
The precise trigger for aPL production is unknown. As with most autoimmune diseases, an interaction between genetic predisposition and environmental triggers is likely.
Genetic factors. APS is not inherited in the strict sense, but certain HLA variants are associated with a higher likelihood of aPL production.
Infectious triggers. Several infections — Epstein–Barr virus, cytomegalovirus, parvovirus B19, certain bacteria — can induce transient aPL in healthy individuals. These typically disappear within months and do not cause thrombosis; only antibodies persisting beyond 12 weeks are considered clinically significant.
Medications. Some drugs (hydralazine, procainamide, chlorpromazine) can induce aPL, but drug-induced APS rarely manifests as thrombosis.
Co-existing autoimmune diseases. Systemic lupus erythematosus, rheumatoid arthritis and Sjögren's syndrome are the most significant risk factors for secondary APS.
Sex and age. APS is substantially more common in women (female-to-male ratio approximately 5:1), predominantly at reproductive age. In men, disease onset is more often associated with arterial rather than venous thrombosis.
Symptoms of Antiphospholipid Syndrome: Thrombosis and Autoimmune Signs
The clinical picture of APS is determined by the site of thrombosis and the degree of organ involvement. The condition can affect virtually any organ.
Venous thrombosis — the most common manifestation. Deep vein thrombosis of the lower limbs followed by pulmonary embolism occurs in approximately half of all patients with thrombotic APS. Recurrence without adequate anticoagulation is characteristic.
Arterial thrombosis — ischaemic stroke and transient ischaemic attacks (TIA) in young adults (under 45–50) without traditional risk factors is one of the key clinical red flags for APS. Myocardial infarction in young people without atherosclerosis, retinal vascular occlusion and limb ischaemia are also well-recognised.
Obstetric complications — covered in detail in the next section.
Thrombocytopenia — a reduction in platelet count, usually mild (typically 50–150 × 10⁹/L). It paradoxically co-exists with thrombosis rather than bleeding, because platelets in APS are hyperactivated.
Livedo reticularis — a characteristic bluish-purple net-like pattern on the skin, particularly on the limbs, due to disrupted microcirculation. Not dangerous in itself, but an important clinical marker of APS.
Cardiac valve disease — vegetations or thickening of the mitral and aortic valves (Libman–Sacks endocarditis) are detected echocardiographically in a proportion of patients.
Renal involvement — renal artery or arteriolar thrombosis, nephrotic syndrome; nephrotic syndrome in APS is less common than in lupus nephritis.
Hypertension in APS is often linked to renal vascular involvement and may respond poorly to standard antihypertensive therapy.
Catastrophic APS (CAPS) — rare (< 1%) but life-threatening: simultaneous thrombosis of small vessels in multiple organs over several days. Mortality without treatment approaches 50%.
APS in Pregnancy: Miscarriage Risk and Obstetric Complications
Obstetric APS is one of the leading causes of recurrent pregnancy loss and severe obstetric complications. Antiphospholipid antibodies damage the placenta through thrombosis of its vessels and direct injury to trophoblast cells.
Criteria for obstetric APS include:
- three or more consecutive miscarriages before 10 weeks of gestation;
- one or more losses of a morphologically normal fetus after 10 weeks;
- premature birth before 34 weeks due to severe pre-eclampsia or placental insufficiency.
Early miscarriages in APS differ from chromosomal losses in that they occur repeatedly and the embryo is morphologically normal. Testing for aPL is mandatory in the investigation of recurrent pregnancy loss.
Without treatment, pregnancy loss rates in APS exceed 90%. With a combination of low-molecular-weight heparin and low-dose aspirin initiated from confirmed pregnancy — or in some cases from the planning stage — live birth rates reach 70–80%. Pregnancy planning in APS requires a joint consultation with a haematologist or rheumatologist and an obstetrician. For monitoring early pregnancy signs in APS, see first signs of pregnancy.
Antiphospholipid Syndrome Diagnosis: Antibodies and Blood Tests That Confirm APS
APS is diagnosed according to the Sydney Criteria (2006) when at least one clinical criterion and at least one laboratory criterion are met, the latter confirmed on two separate occasions at least 12 weeks apart.
Laboratory criteria — three antibody groups:
- Lupus anticoagulant (LA) — a functional assay: antibodies prolong phospholipid-dependent coagulation tests in vitro. Detected through coagulation testing (aPTT, dRVVT). The paradox: an "anticoagulant" in the test tube is a powerful procoagulant in the body.
- Anticardiolipin antibodies (aCL) IgG and IgM — ELISA; diagnostically significant at medium and high titres (> 40 GPL or MPL).
- Anti-beta-2-glycoprotein I antibodies (anti-β₂GPI) IgG and IgM — the most specific marker for true APS.
Positivity for all three antibody groups ("triple positivity") marks the highest thrombotic risk profile.
Additional investigations:
- D-dimer — elevated in active thrombosis; used to confirm a thrombotic event and monitor treatment response.
- Full blood count — thrombocytopenia as a manifestation of APS.
- Antinuclear antibodies (ANA), anti-dsDNA — to exclude co-existing SLE.
- Kidney function tests — to assess renal involvement.
A fundamental rule: a positive aPL test on first testing is not a diagnosis. Mandatory confirmation ≥ 12 weeks later is required. Transient aPL associated with infections are clinically insignificant.
Treatment of Antiphospholipid Syndrome
APS treatment aims to prevent recurrent thrombosis and obstetric complications — current medicine cannot eliminate the autoimmune process or eradicate the antibodies.
For thrombotic APS:
- Warfarin — historically the primary agent for long-term secondary thrombosis prevention in APS. Target INR 2.0–3.0 for venous thrombosis; for arterial thrombosis — often 3.0–4.0. Requires regular laboratory monitoring.
- Direct oral anticoagulants (DOACs) (rivaroxaban, apixaban) — less effective in "triple-positive" patients based on clinical trial data; used only in low-risk profiles or when warfarin is not feasible.
- Hydroxychloroquine — an immunomodulator with antithrombotic properties; recommended for all APS patients, particularly those with co-existing SLE.
- Statins and low-dose aspirin — added when cardiovascular risk is high.
Duration of therapy: with "triple positivity" or an arterial thrombotic event, most guidelines support lifelong anticoagulation; with isolated antibody positivity and a first venous event, a shorter course may be considered after careful individual risk assessment.
For obstetric APS (no prior thrombosis):
- Combination of low-molecular-weight heparin (LMWH) + low-dose aspirin (75–100 mg/day) from confirmed pregnancy — or from the planning stage in some cases — through to 34–36 weeks, then aspirin alone.
- Hydroxychloroquine or intravenous immunoglobulin are added when the standard regimen fails.
For catastrophic APS — intensive care: high-dose heparin anticoagulation, glucocorticoids, plasmapheresis, intravenous immunoglobulin.
When to See a Doctor
Mandatory rheumatology or haematology referral when:
- thrombosis at any site in a person under 50 without traditional risk factors;
- stroke or TIA in a young adult without hypertension or atherosclerosis;
- two or more consecutive miscarriages or one pregnancy loss after 10 weeks;
- combination of thrombocytopenia and thrombotic events;
- characteristic livedo reticularis combined with any of the above.
Testing positive for aPL without clinical events — "aPL carriership" — is not a diagnosis of APS and does not require anticoagulation, but does call for elimination of additional thrombotic risk factors. Do not interpret test results independently — management is determined by a physician.
This content is for informational purposes only and does not replace professional medical advice.
Frequently Asked Questions
Lupus anticoagulant (LA) is an antiphospholipid antibody that prolongs phospholipid-dependent clotting tests in vitro — hence 'anticoagulant'. It was first described in patients with systemic lupus erythematosus — hence 'lupus'. Today it is known to occur in primary APS without lupus as well. The paradox: despite prolonging test tube clotting times, this 'anticoagulant' is a powerful procoagulant in the living body and substantially raises thrombosis risk.
Isolated aPL positivity without clinical events is not APS. Prophylactic anticoagulation is not indicated in this setting because bleeding risk outweighs benefit. The recommended approach is to eliminate additional thrombotic risk factors: smoking cessation, control of hypertension, weight normalisation, caution with hormonal contraceptives. During pregnancy, aPL carriership requires specialist monitoring even without a prior thrombotic event.
Duration depends on the type of event and the antibody profile. With 'triple positivity' or an arterial thrombotic event, most guidelines recommend lifelong anticoagulation. For a first venous thrombosis with single antibody positivity, a shorter treatment course may be appropriate — but only after an individual risk-benefit assessment with a haematologist. Self-discontinuing anticoagulants in APS is dangerous — recurrence risk is very high.
APS and elevated homocysteine are two separate thrombotic risk factors that can co-exist in the same patient and multiplicatively amplify clot risk. When investigating thrombophilia, homocysteine is checked alongside antiphospholipid antibodies — it is a standard component of the complete thrombophilia screen.
APS is not a classical hereditary disease. However, certain genetic variants increase susceptibility to aPL production. First-degree relatives of APS patients have a modestly elevated risk of developing an autoimmune condition, but screening asymptomatic relatives is not standard practice in the absence of symptoms.
The defining differences are APS's autoimmune and acquired nature, while most other thrombophilias (protein C deficiency, factor V Leiden) are inherited. APS involves both venous and arterial thrombosis — most hereditary thrombophilias are confined to venous events. APS is unique in causing obstetric complications and systemic autoimmune features (livedo, thrombocytopenia). In APS, D-dimer is used not only to confirm acute thrombosis at presentation, but also to monitor anticoagulation response over time.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service