Homocysteine Blood Test: Normal Levels, Causes and Results
A homocysteine test is ordered when investigating an unexplained thrombosis in a young person, during pre-conception screening or after a stroke without obvious risk factors. But what does this marker actually measure — and why does its level matter so much for vascular health? Homocysteine is an amino acid that the body normally clears rapidly, but when B-vitamin metabolism is disrupted it accumulates and begins to chemically erode arterial walls from within.
What Is Homocysteine and Why Is It Measured?
Homocysteine is a sulphur-containing amino acid and an intermediate product of methionine metabolism. The body does not obtain it from food directly: it is synthesised from methionine and must be rapidly recycled — either back into methionine (remethylation, requiring vitamin B12 and folate) or converted into cysteine (transsulphuration, requiring vitamin B6).
When either pathway is impaired — due to nutrient deficiency, genetic variants or concurrent disease — homocysteine accumulates in the blood. Elevated levels damage endothelial cells, the thin inner lining of arteries: the wall loses its protective properties, becomes permeable to lipids, and the risk of thrombus formation and atherosclerosis rises substantially.
Homocysteine is measured to assess cardiovascular risk, investigate the cause of thrombotic events, evaluate B-vitamin metabolism disorders and screen patients planning a pregnancy.
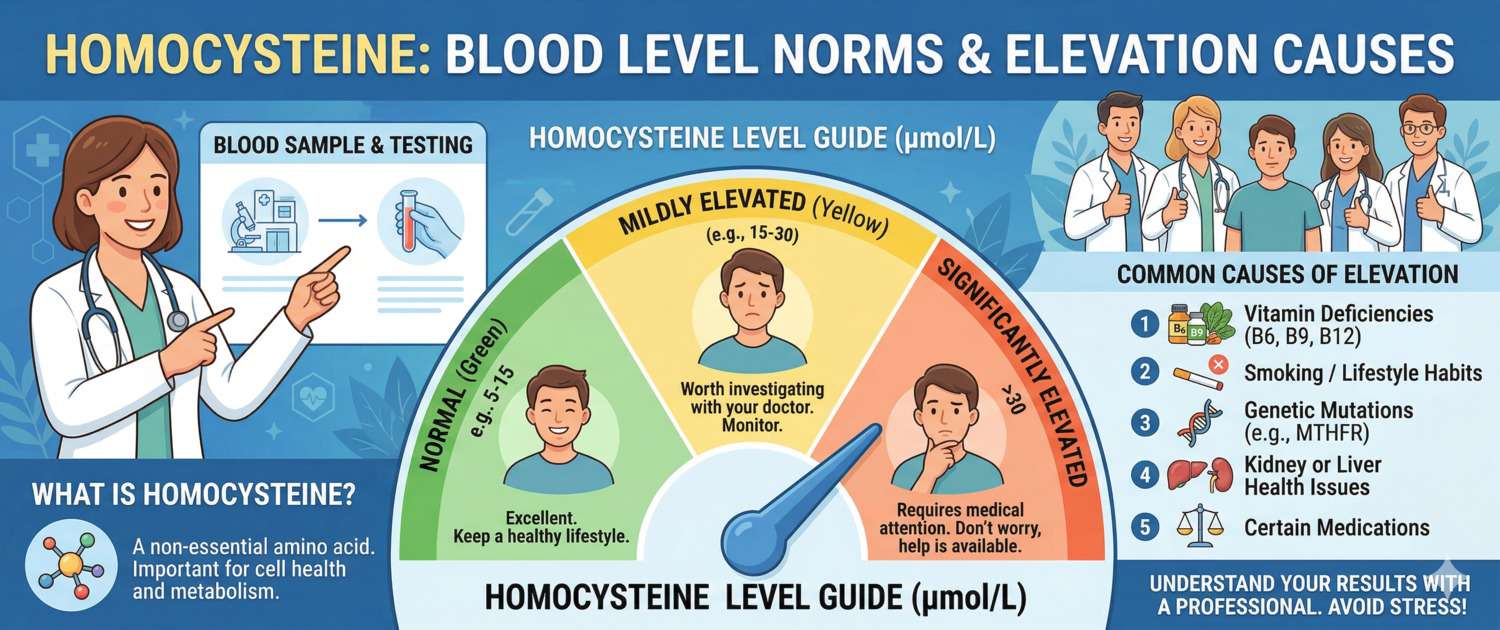
Homocysteine Normal Ranges for Adults
Reference values depend on age and sex. Levels rise physiologically with age, partly because kidney function — a key clearance route — declines over time.
| Group | Normal range (µmol/L) |
|---|---|
| Women 18–45 years | 5–12 |
| Men 18–45 years | 6–15 |
| Women > 45 years | 5–15 |
| Men > 45 years | 6–20 |
| Pregnant women (first trimester) | < 8 |
Elevated homocysteine — hyperhomocysteinaemia — is graded as mild (15–30 µmol/L), moderate (31–100 µmol/L) or severe (> 100 µmol/L). The severe form is rare and almost always indicates a hereditary disorder called homocystinuria.
In pregnancy, thresholds are tighter: levels above 8–10 µmol/L in the first trimester are associated with increased risk of fetal neural tube defects, pre-eclampsia and miscarriage.
Causes of Elevated Homocysteine
Homocysteine accumulation is nearly always a consequence of impaired clearance. Several causes often act together.
B-vitamin deficiency is the most common and most correctable cause. Vitamin B12 deficiency blocks the remethylation of homocysteine back to methionine: without the cofactor the reaction stalls and the amino acid builds up. Folate deficiency acts through the same mechanism — folate donates the methyl group needed for this reaction. Vitamin B6 deficiency disrupts the alternative clearance route, transsulphuration.
MTHFR gene polymorphism. Mutations in the methylenetetrahydrofolate reductase gene reduce the activity of the enzyme involved in remethylation. The C677T variant is present in 10–15% of Europeans; the homozygous form causes a moderate homocysteine rise, especially when folate intake is also low.
Kidney disease. The kidneys actively metabolise homocysteine, and as glomerular filtration rate falls, plasma levels rise in proportion.
Medications. Methotrexate blocks folate metabolism. Metformin and proton pump inhibitors reduce vitamin B12 absorption. Smoking, excessive alcohol consumption and physical inactivity are additional contributing factors.
How High Homocysteine Damages Blood Vessels
Homocysteine is not merely a passive risk marker — it actively injures vascular walls. The process resembles acid running through metal pipes: the exterior looks intact while the interior corrodes progressively.
Specifically, elevated homocysteine oxidises endothelial cells, enhances platelet adhesion, activates the coagulation cascade and creates a prothrombotic environment. This is directly linked to the risk of deep vein thrombosis and arterial thrombosis — particularly in young adults without conventional risk factors. In thrombophilia workups, homocysteine is a standard test alongside D-dimer and genetic coagulation markers.
Over the long term, even mild hyperhomocysteinaemia approximately doubles the risk of myocardial infarction and raises ischaemic stroke risk by 40–50%. The association with heart attack is especially pronounced in patients lacking traditional risk factors, which is precisely when homocysteine testing often reveals the hidden cause.
Symptoms of Hyperhomocysteinaemia
Mild elevation — 15–30 µmol/L — is entirely asymptomatic. The patient feels nothing unusual until a thrombotic or vascular event occurs. This is why testing is based on risk factors rather than symptoms.
In severe hyperhomocysteinaemia (homocystinuria, > 100 µmol/L), clinical features are striking: ectopia lentis (lens dislocation), skeletal deformities, early-onset atherosclerosis, thrombosis in childhood and adolescence, and cognitive developmental delay.
How to Prepare for a Homocysteine Blood Test
Homocysteine is one of the markers where pre-analytical preparation is critically important. Failure to follow the rules can inflate the result by 50–100%.
Strict fasting for 12 hours: protein-rich food — especially methionine-rich sources such as meat and eggs — sharply raises plasma homocysteine. No alcohol for 24 hours — alcohol disrupts folate metabolism and distorts the result. Cold chain is essential: after collection the tube must be immediately placed on ice and delivered to the laboratory within 30 minutes — in a warm sample, red blood cells continue to release homocysteine, falsely elevating the reading. Inform your doctor about methotrexate, metformin or proton pump inhibitors — all require consideration when interpreting results.
How to Lower Homocysteine: The Role of B Vitamins
When mild-to-moderate hyperhomocysteinaemia is caused by B-vitamin deficiency, correction is well studied and effective.
Folic acid 0.4–5 mg/day lowers homocysteine by approximately 25%. Adding vitamin B12 produces a further 7% reduction and is essential when B12 deficiency is confirmed. Vitamin B6 is added when transsulphuration dysfunction is suspected. Combined supplementation with all three B vitamins is the standard approach when elevation is documented.
An important nuance: lowering homocysteine with vitamins reliably reduces stroke recurrence — but not myocardial infarction, according to large randomised trials. This means vitamin correction addresses one component of cardiovascular risk, not the whole picture.
When High Homocysteine Requires Urgent Evaluation
A routine follow-up appointment is appropriate for mild elevations found on screening. Certain situations warrant prompt investigation:
- Deep vein thrombosis or pulmonary embolism under the age of 45 without an obvious provoking factor;
- Stroke or heart attack in a person with no conventional risk factors;
- Homocysteine above 50 µmol/L on first measurement;
- Recurrent miscarriage or pre-eclampsia.
In these scenarios, hyperhomocysteinaemia may be part of a broader coagulation disorder requiring full haematological investigation.
Homocysteine is included in the vascular ageing and biological age marker panel — see the article biological age by blood tests. For longevity-optimal homocysteine targets, the B-vitamin reduction protocol and the dementia risk link, see the article homocysteine and longevity.
Frequently Asked Questions
Elevated homocysteine indicates impaired clearance — most commonly due to vitamin B12, B6 or folate deficiency, and less often due to genetic variants or kidney disease. High levels damage arterial walls and create conditions for clot formation, significantly increasing the risk of deep vein thrombosis and heart attack — especially in young adults with no other obvious risk factors.
In women of reproductive age the normal range is 5–12 µmol/L; in men of the same age it is 6–15 µmol/L. After age 45 the upper limit rises slightly — to 15 µmol/L in women and 20 µmol/L in men. In the first trimester of pregnancy, the target is below 8 µmol/L, as higher levels are associated with fetal risk.
The most extreme rises — above 100 µmol/L — occur in hereditary homocystinuria and severe chronic kidney failure. Moderate elevation to 30–50 µmol/L is characteristic of vitamin B12 deficiency, severe folate deficiency and thrombophilia. When a coagulation disorder is suspected, homocysteine is part of the standard diagnostic panel.
When B-vitamin deficiency is the cause, folic acid combined with vitamins B12 and B6 lowers homocysteine by 25–35%. Supporting measures include quitting smoking, limiting alcohol and maintaining regular moderate physical activity. Dietary changes — increasing leafy greens, legumes and fortified grains — add further benefit. Self-supplementation without a test is not advisable: it is important to establish the underlying cause first.
Yes — preparation matters considerably here. Blood must be drawn after at least 12 hours of fasting: a protein-rich dinner the night before can distort results. After collection, the tube must be immediately cooled and delivered to the laboratory within 30 minutes, as homocysteine continues to rise in warm samples. Inform your doctor about any medications, particularly metformin, proton pump inhibitors or methotrexate.
Yes. Mild hyperhomocysteinaemia approximately doubles the risk of myocardial infarction and raises ischaemic stroke risk by around 40–50%. Vitamin B correction reliably reduces stroke recurrence but has less clear benefit for heart attack prevention, making homocysteine management one part of a broader cardiovascular risk strategy.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service