Hyperuricaemia: Causes, Symptoms and How to Lower Uric Acid
A blood test reveals elevated uric acid, but joints are fine and gout has never been diagnosed. Should you be concerned? Yes — but calmly. Hyperuricaemia — persistently elevated uric acid — can exist for years without any symptoms while silently damaging the kidneys, blood vessels, and joints. Here is where uric acid comes from, when it becomes genuinely dangerous, and what to do about it.
What Uric Acid Is and Where It Comes From
Uric acid is the end product of purine metabolism — purines are the nitrogen bases that make up DNA. When cells die or food is digested, purines are broken down to uric acid. It is excreted primarily by the kidneys (approximately 70%) and through the gut (30%).
Under normal conditions, uric acid production and elimination are balanced. Hyperuricaemia develops when this balance fails: either too much is produced (accelerated cell turnover, excess dietary purines) or the kidneys excrete too little. Usually both mechanisms act together.
Uric acid is not simply metabolic waste — at physiological concentrations it has antioxidant properties. The problem begins when its concentration exceeds the solubility threshold: urates crystallise in tissues, joints, and kidneys.
Normal Uric Acid Levels
Reference ranges depend on sex and, in women, menopausal status — oestrogens enhance renal urate excretion.
| Group | Normal (µmol/L) | Normal (mg/dL) |
|---|---|---|
| Men | 200–420 | 3.4–7.0 |
| Pre-menopausal women | 140–360 | 2.4–6.0 |
| Post-menopausal women | 200–420 | 3.4–7.0 |
Hyperuricaemia is defined as above 420 µmol/L (7.0 mg/dL) in men and above 360 µmol/L (6.0 mg/dL) in pre-menopausal women. The crystallisation threshold for urates is approximately 420 µmol/L at normal body temperature.
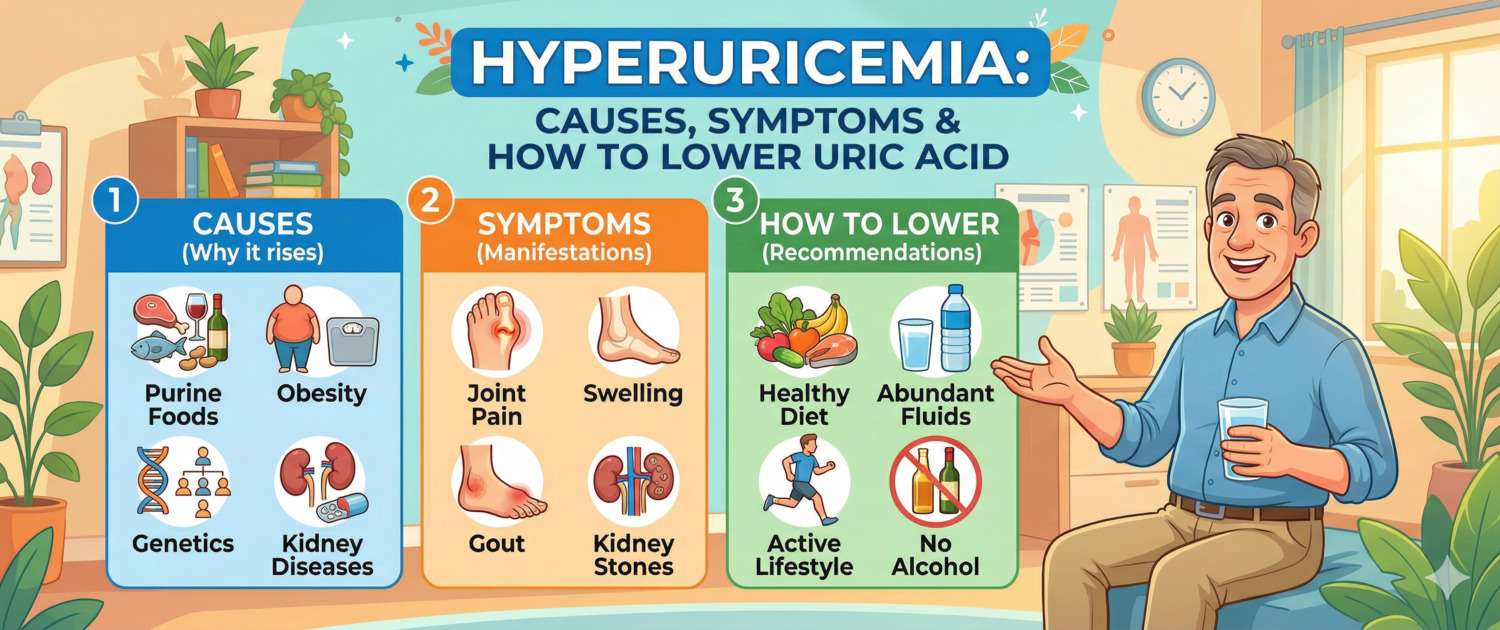
Causes of Hyperuricaemia
Primary hyperuricaemia — genetically determined reduction in renal urate excretion or overproduction (rare enzyme defects, such as HPRT deficiency — Lesch-Nyhan syndrome).
Secondary hyperuricaemia — a consequence of another condition or exposure:
- Purine-rich diet: red meat, organ meats (liver, kidneys), seafood (anchovies, sardines, mussels), meat broths, yeast. Alcohol — especially beer and spirits — raises uric acid through a dual mechanism: purines in beer plus impaired renal excretion from ethanol.
- Renal failure — reduced urate clearance. Rising creatinine and uric acid parallel each other in chronic kidney disease.
- Metabolic syndrome and insulin resistance — insulin reduces renal urate excretion; obesity additionally increases production.
- Medications: thiazide diuretics and furosemide are the most common iatrogenic causes. Low-dose aspirin, ciclosporin, and nicotinic acid also raise uric acid.
- Cancer and its treatment: massive tumour cell breakdown (tumour lysis syndrome during chemotherapy) releases a surge of purines. Multiple myeloma and other haematological malignancies are typical causes of secondary hyperuricaemia.
- Hypothyroidism — reduces renal urate clearance.
Why Asymptomatic Hyperuricaemia Is Dangerous
Many people with hyperuricaemia have never had a gout attack and regard elevated uric acid as a harmless incidental finding. This is a misconception — chronic hyperuricaemia damages organs even without visible symptoms.
Gout is not the only consequence of hyperuricaemia — it is just the most dramatic one. Most people with elevated uric acid go years without a gout attack, but silent organ damage is already under way. Full details are in the gout article.
Urate nephropathy — deposition of urate crystals in the renal tubules and interstitium. Manifests as reduced urinary concentrating ability, proteinuria, and a gradual rise in creatinine. Prolonged severe hyperuricaemia can lead to chronic kidney disease.
Nephrolithiasis — urate stones account for 5–10% of all kidney stones. Stone formation risk rises sharply in acidic urine (pH < 5.5) — the pH at which urates transition from dissolved to crystalline form.
Cardiovascular risk — hyperuricaemia is associated with elevated risks of hypertension, metabolic syndrome, and cardiovascular events, though whether it is an independent causal factor remains actively debated.
Diagnostic Approach
Uric acid in blood — the primary test. Drawn fasting; alcohol, purine-rich food, and intense exercise are avoided for 24 hours beforehand, as all transiently raise levels. A single elevated result is always repeated.
Creatinine and eGFR — renal function assessment. In hyperuricaemia secondary to renal failure, cause and effect are closely intertwined.
Urinalysis — uric acid excretion and urine pH are assessed. Persistently acidic urine (pH < 5.5) is a risk factor for urate nephrolithiasis. A 24-hour uric acid excretion above 800 mg/day indicates overproduction; a low excretion points to impaired renal clearance.
Kidney function test — comprehensive renal assessment when urate nephropathy is suspected.
Treatment and Uric Acid Reduction
Strategy depends on the uric acid level, the presence of symptoms, and comorbidities.
Diet and Lifestyle — the First Step
Restrict or eliminate: red meat and organ meats; high-purine seafood (anchovies, sardines, mussels, oysters); alcohol — especially beer; fructose-containing sweet drinks (fructose accelerates uric acid synthesis).
Increase: water intake (2–3 L/day) — alkalinises urine and reduces crystallisation risk; low-fat dairy products — associated with lower uricaemia; coffee — moderate consumption is linked to lower uric acid levels.
Dietary measures reduce uric acid by approximately 60–90 µmol/L — meaningful, but often insufficient to normalise levels in significant hyperuricaemia.
Drug Treatment
Indications for drug therapy in asymptomatic hyperuricaemia are not universally agreed. Most guidelines recommend treatment when:
- Gouty arthritis (any episode) has occurred
- Urate nephrolithiasis is present
- Urate nephropathy is established
- Tophaceous gout is present
- Uric acid exceeds 540 µmol/L in patients with high cardiovascular risk or chronic kidney disease
Allopurinol — xanthine oxidase inhibitor, reduces uric acid synthesis. First-line treatment for most indications. Started at a low dose (100 mg/day) with gradual titration to reach the target level (< 360 µmol/L).
Febuxostat — alternative to allopurinol when the latter is not tolerated or in renal impairment.
Probenecid, benzbromarone — uricosuric agents (enhance renal excretion). Used when excretion is low and kidney function is preserved.
When to See a Doctor
Planned rheumatology or GP visit: uric acid above 480 µmol/L under any circumstances; hyperuricaemia combined with impaired kidney function or kidney stones; recurrent episodes of joint pain after heavy meals or alcohol.
Urgently: acute arthritis with redness, swelling, and excruciating pain in a joint (especially the big toe) — the first sign of gout requiring immediate treatment.
For male-specific uric acid reference ranges by age and the link with cardiovascular risk, see the article uric acid in men.
This article is for informational purposes only. Diagnosis and treatment are determined by a rheumatologist or general physician.
Frequently Asked Questions
Hyperuricaemia is a laboratory finding: uric acid elevated above 420 µmol/L in men or 360 µmol/L in pre-menopausal women. Gout is a clinical disease: acute arthritis caused by urate crystals depositing in a joint. Most people with hyperuricaemia never experience a gout attack, but the risk is directly proportional to the uric acid level and how long it has been elevated.
It depends on the uric acid level and comorbidities. With moderate elevation (420–480 µmol/L) and no evidence of kidney or joint damage, dietary modification and monitoring are sufficient. With significant elevation (> 540 µmol/L), especially combined with reduced kidney function, urate stones, or high cardiovascular risk, drug treatment is justified. The decision is made by a physician.
The greatest contributors are organ meats (liver, kidneys, brain), red meat, high-purine seafood (anchovies, sardines, mussels, oysters), meat broths, and yeast. Alcohol — especially beer — raises uric acid through two mechanisms: it contains purines and impairs renal excretion. Fructose in sweet carbonated drinks accelerates uric acid synthesis.
Yes. Chronic hyperuricaemia causes urate nephropathy — crystal deposition in the renal tubules — independently of gout attacks. This leads to gradually declining kidney function, detected by rising creatinine and falling eGFR. Prolonged severe hyperuricaemia can eventually cause chronic kidney disease.
Yes — weight loss reduces uric acid through several mechanisms: improved insulin sensitivity, lower urate production with reduced cell mass, and decreased inflammatory load. However, very rapid weight loss (fasting, aggressive diets) can transiently raise uric acid due to tissue breakdown — which is why weight reduction should be gradual and steady.
The basic panel: uric acid repeated fasting, creatinine with eGFR calculation, a full kidney function test, and urinalysis with pH and uric acid excretion. When risk factors are present, assessment for metabolic syndrome is added: glucose, lipid panel, and blood pressure.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service