Insulin Resistance: Causes, Symptoms and How to Diagnose
Excess weight around the abdomen, persistent fatigue after meals, and a craving for sugar that feels impossible to control — behind these seemingly ordinary complaints can lie insulin resistance. Estimates suggest it affects 30–40% of adults in developed countries, yet most people are unaware of it until prediabetes or metabolic syndrome has already developed. Here is what it is, how to recognise it, and what to do.
What Is Insulin Resistance and How Does It Develop
Insulin is a pancreatic hormone whose main job is to "open the door" into cells for glucose. Normally, after a meal blood glucose rises, the pancreas releases insulin, it binds to receptors on muscle, liver, and fat cells — and glucose enters, providing energy or being stored.
In insulin resistance, cells stop responding normally to insulin. Think of a rusted lock: the right key is inserted, but the door will not open. The pancreas "sees" that glucose is not clearing from the blood and begins releasing more and more insulin to overcome the resistance. This works — but only for a while. As the pancreatic reserve is exhausted, prediabetes develops, and eventually type 2 diabetes.
Chronically elevated insulin is itself harmful: it stimulates fat deposition (especially visceral fat around internal organs), suppresses fat breakdown, drives androgen production in the ovaries, and activates the sympathetic nervous system — raising blood pressure. This is how insulin resistance becomes the engine driving several chronic diseases simultaneously.
Causes and Risk Factors for Insulin Resistance
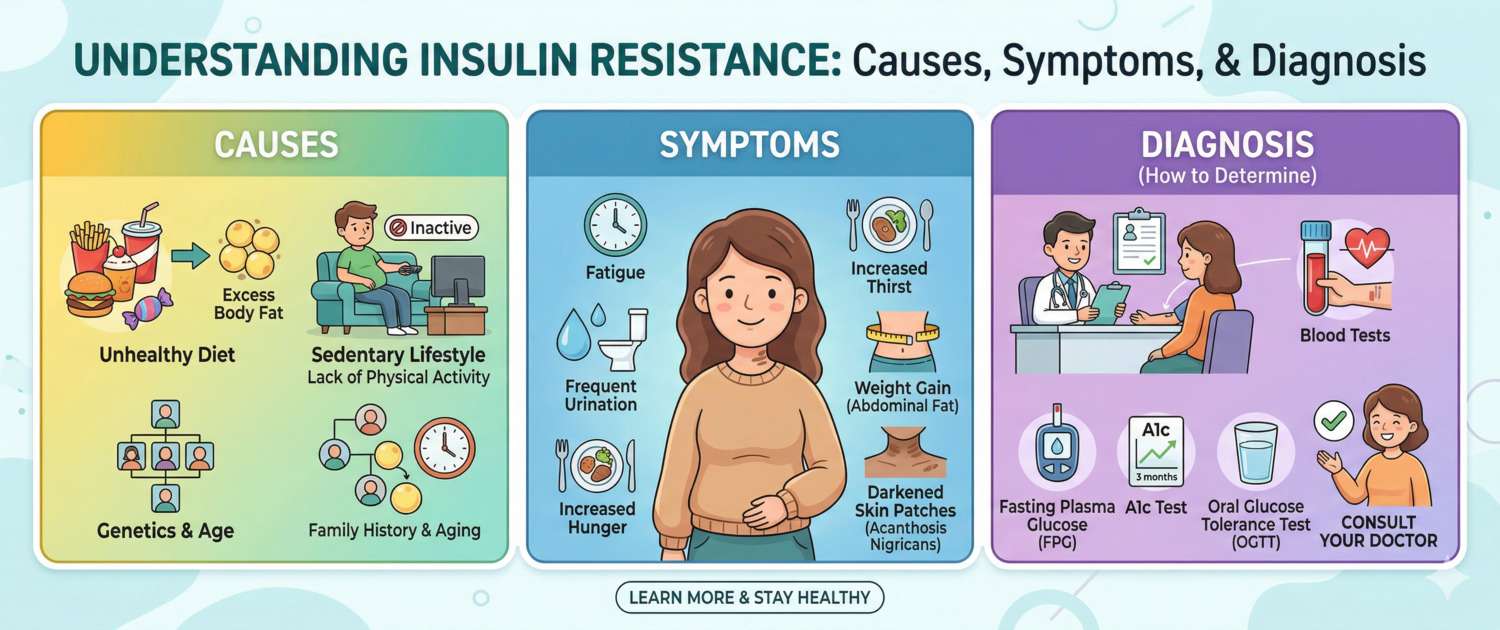
Obesity, especially visceral. Abdominal fat is metabolically active: it releases inflammatory cytokines and free fatty acids that disrupt insulin signalling in muscle and liver cells. This is the primary cause in the majority of patients.
Physical inactivity. Muscle is the body's main consumer of glucose. With low activity, muscle mass falls, the number of insulin receptors decreases, and insulin sensitivity drops. Just a few days of bed rest in a healthy person reduces insulin sensitivity by 25–40%.
Poor sleep and chronic stress. Sleeping fewer than 6 hours per night raises cortisol and reduces insulin sensitivity within 1–2 weeks. Chronic stress acts through the same mechanism.
High glycaemic load diet. A diet rich in refined carbohydrates and sugar creates frequent glucose spikes — the pancreas constantly operates under high load, and receptors gradually "fatigue" their response.
Genetic predisposition. Insulin resistance has a strong familial component: if parents had type 2 diabetes, personal risk is significantly elevated.
Hormonal conditions. Polycystic ovary syndrome (PCOS) — approximately 70% of women with this diagnosis have insulin resistance. Hypothyroidism, Cushing's syndrome, and hyperprolactinaemia also worsen insulin sensitivity.
Ethnicity. People of South Asian, Latin American, and Middle Eastern descent are genetically more prone to insulin resistance at the same BMI as Europeans.
Symptoms of Insulin Resistance: How to Suspect the Condition
Insulin resistance is asymptomatic for a long time. But a cluster of indirect signs appears frequently enough to be worth knowing.
Metabolic signs:
- weight gain predominantly in the abdomen (waist circumference > 94 cm in men, > 80 cm in women);
- difficulty losing weight even when calories are restricted;
- pronounced fatigue and drowsiness 1–2 hours after a carbohydrate-rich meal.
Skin signs:
- acanthosis nigricans — darkening and thickening of skin in body folds (neck, armpits, groin, elbows). This is a direct result of chronically elevated insulin stimulating keratinocyte proliferation;
- multiple small skin tags (acrochordons) on the neck and in the armpits.
Other signs:
- cravings for sugar and carbohydrates that intensify in the afternoon;
- difficulty concentrating, "brain fog";
- in women — irregular periods, excess hair growth (hirsutism), acne — signs of hyperandrogenism driven by high insulin acting on ovarian cells.
None of these signs is specific to insulin resistance. Diagnosis is established through blood tests, not symptoms alone.
Tests and HOMA-IR Index: How to Diagnose Insulin Resistance
There is no universally standardised clinical test for insulin resistance — the "gold standard" (euglycaemic hyperinsulinaemic clamp) is used only in research. In clinical practice, indirect markers are used.
HOMA-IR index — the most widely used calculated measure:
HOMA-IR = (fasting glucose, mmol/L × fasting insulin, µIU/mL) ÷ 22.5
A value above 2.7 (in some laboratories above 2.5) indicates insulin resistance. Critically: both fasting glucose and fasting insulin must be drawn strictly after an 8–12 hour fast — otherwise the result is unreliable.
Glycated haemoglobin (HbA1c) — reflects average glucose over the past 2–3 months. In insulin resistance it is often in the upper normal range (5.5–5.9%) or already in the prediabetes range (6.0–6.4%).
Lipid profile: the characteristic pattern in insulin resistance is elevated triglycerides (> 1.7 mmol/L) and reduced HDL cholesterol (< 1.0 in men, < 1.3 in women). This combination with visceral obesity and hypertension constitutes the full metabolic syndrome.
Oral glucose tolerance test — a more sensitive approach: glucose is measured fasting and again 2 hours after ingesting 75 g of glucose. A 2-hour value of 7.8–11.0 mmol/L indicates impaired glucose tolerance — the intermediate state between insulin resistance and overt diabetes.
Insulin Resistance and Associated Conditions
Insulin resistance rarely exists in isolation — it is the pathophysiological foundation for a cluster of interconnected conditions.
Type 2 diabetes — the direct downstream consequence of progressive insulin resistance. As beta-cell capacity is exhausted, glucose can no longer be controlled even by high insulin levels — overt diabetes develops. Insulin resistance precedes diabetes by years or decades: this is the best window for prevention.
Arterial hypertension — chronically elevated insulin activates the sympathetic nervous system and promotes renal sodium retention. Approximately 50% of patients with hypertension have co-existing insulin resistance.
Non-alcoholic fatty liver disease (NAFLD) — excessive free fatty acid influx and enhanced hepatic lipogenesis driven by high insulin lead to fat accumulation in hepatocytes. NAFLD and insulin resistance mutually reinforce each other.
Polycystic ovary syndrome — high insulin stimulates excessive androgen production in the ovaries, disrupting follicle maturation. This is one of the core mechanisms of PCOS, explaining why metformin (an insulin-sensitising drug) is effective in this condition.
Cardiovascular disease — the dyslipidaemia, hypertension, and chronic inflammation accompanying insulin resistance accelerate atherosclerosis and elevate myocardial infarction risk independently of glucose levels.
Reversing Insulin Resistance: Diet, Activity and Medication
The good news: insulin resistance is reversible — especially in its early stages. Lifestyle change outperforms any single medication.
Physical activity is the most powerful tool. Resistance training increases muscle mass and insulin receptor density; aerobic exercise reduces visceral fat and systemic inflammation. Even 150 minutes of moderate aerobic activity per week reduces HOMA-IR by 25–30% over 3 months.
Nutrition. No single "correct" diet exists, but evidence-backed principles include:
- prioritising whole foods with a low glycaemic load (vegetables, legumes, wholegrains, lean proteins);
- reducing refined carbohydrates, sugar-sweetened beverages, and trans fats;
- modest caloric deficit: a 5–7% reduction in body weight is sufficient to meaningfully improve insulin sensitivity;
- the Mediterranean or DASH dietary pattern — the most studied and effective models for insulin resistance.
Sleep normalisation. At least 7 hours per night is a non-negotiable baseline. Chronic sleep deprivation negates even optimal diet and exercise in terms of insulin sensitivity.
Pharmacological therapy. When insulin resistance is confirmed alongside glucose dysregulation, metformin is prescribed — it reduces hepatic glucose output and improves peripheral tissue sensitivity. In PCOS, it additionally lowers androgen levels. GLP-1 receptor agonists (semaglutide, liraglutide) produce significant weight loss and improve insulin sensitivity — indicated in obesity and prediabetes.
When to See a Doctor
See an endocrinologist or general practitioner if several of the following apply:
- waist circumference above threshold combined with post-meal fatigue and sugar cravings;
- darkening of skin folds on the neck or in the armpits;
- irregular periods in women without another identified cause;
- fasting glucose 5.6–6.9 mmol/L on more than one test;
- HbA1c in the 5.7–6.4% range;
- triglycerides above 1.7 mmol/L and low HDL;
- a first-degree relative with type 2 diabetes.
Insulin resistance is not a verdict — it is a signal caught early enough to change the trajectory and prevent diabetes, myocardial infarction, and other downstream consequences. Do not self-diagnose based on symptoms and an online calculator — see a doctor for a full workup.
Frequently Asked Questions
The primary method is the HOMA-IR index, calculated as (fasting glucose × fasting insulin) ÷ 22.5. A value above 2.7 indicates insulin resistance. Both measurements must be taken after a strict 8–12 hour fast. Additionally, HbA1c, triglycerides, and HDL cholesterol are assessed — the characteristic pattern helps confirm the diagnosis.
Yes, at early stages lifestyle change is more effective than medication. 150 minutes of aerobic activity per week, 5–7% weight loss, and reducing refined carbohydrates lower HOMA-IR by 25–40% over 3 months. Sleep normalisation — a minimum of 7 hours per night — is essential: chronic sleep deprivation negates even a perfect diet.
Directly and bidirectionally. Visceral fat releases inflammatory substances that block insulin signalling, leading to insulin resistance. In turn, chronically high insulin promotes fat deposition and makes weight loss harder. This vicious cycle is most effectively broken by physical activity and a modest caloric deficit.
Yes. So-called metabolically unhealthy normal weight occurs in 10–15% of people with a normal BMI, particularly those with low muscle mass and high visceral fat (thin limbs, protruding abdomen). Such individuals carry all the metabolic risks of obesity without appearing overweight. Waist circumference is the best simple indicator of visceral fat.
Insulin resistance disrupts lipid metabolism: the liver synthesises more triglycerides and LDL cholesterol, while HDL cholesterol falls. This dyslipidaemia accelerates atherosclerosis independently of glucose levels. This is why a full lipid panel is always checked when insulin resistance is suspected or confirmed.
Yes, through hyperandrogenism. High insulin stimulates the ovaries to overproduce androgens, disrupting follicle maturation and causing anovulation. This is the central mechanism in PCOS — the leading cause of anovulatory infertility. Correcting insulin resistance through metformin, weight loss, and exercise often restores ovulation and fertility.
Upload your lab results photo or PDF
AI explains your results in 30 seconds
Choose fileRate the service
Your feedback helps us improve the service